Identification of a miRNA signature for schizophrenia in plasma-derived extracellular vesicles
Identification of a miRNA signature for schizophrenia in plasma-derived extracellular vesicles
Collier, M. E. W.; Chiappelli, J.; Marshall, H.; Sylvius, N.; Allcock, N.; Whittingham, J.; Kochunov, P.; Schwarcz, R.; Hong, E. L.; Giorgini, F.
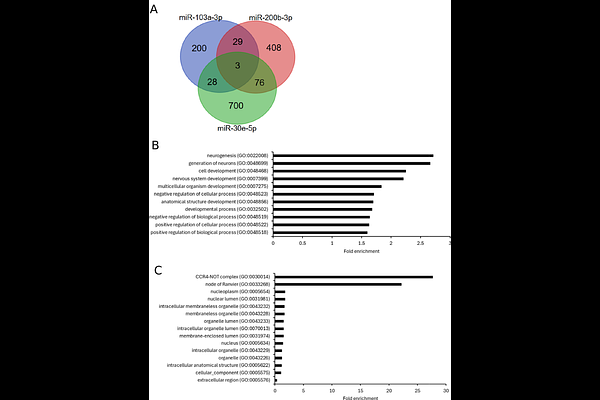
AbstractBackground and Hypothesis: Extracellular vesicles (EVs) are phospholipid bilayer vesicles released from cells containing proteins, lipids and nucleic acids derived from the parent cell. Alterations in miRNA expression within blood-derived EVs have been proposed as potential biomarkers of disease. Specifically, identification of differentially expressed miRNAs in patients with schizophrenia (SZ) compared to healthy individuals could be used as a miRNA signature to aid in diagnosis and treatment. We therefore aimed to identify differentially expressed miRNAs in plasma-derived EVs between people living with SZ and healthy controls and to correlate miRNA levels with SZ-relevant clinical measures. Study Design: Plasma-derived EVs were isolated from a cohort of 33 individuals with SZ and 34 controls. Expression of 84 miRNAs was examined using a RT-qPCR panel. Study Results: Three miRNAs (hsa-miR-30e-5p, hsa-miR-103a-3p, hsa-miR-200b-3p) were differentially expressed between controls and patients. Gene ontology analysis of putative target genes shared between these miRNAs revealed enrichment of biological process terms related to neurogenesis. Analysis of miRNA expression compared to clinical measures showed that hsa-miR-103a-3p expression was associated with working memory and negatively correlated with white matter integrity in the combined patient-control group. Conclusions: We have identified a miRNA signature for SZ in plasma-derived EVs and shown for the first time that hsa-miR-30e-5p expression is significantly increased in plasma-derived EVs in SZ. The genetic links between differentially expressed miRNAs and neurogenesis, along with the correlations of hsa-miR-103a-3p with working memory and white matter integrity may underlie the functional importance of altered expression of the identified miRNAs in SZ.