Female-enriched Eggerthella lenta drives neuroinflammation and IFN-γ via host receptor TLR2
Female-enriched Eggerthella lenta drives neuroinflammation and IFN-γ via host receptor TLR2
Rock, R. R.; Alexander, M.; Noecker, C.; Trepka, K.; Upadhyay, V.; Ortega, E.; Ramirez, L.; Siewart, L.; Olson, C.; Halsey, T.; Probstel, A.-K.; Baranzini, S.; Turnbaugh, P. J.
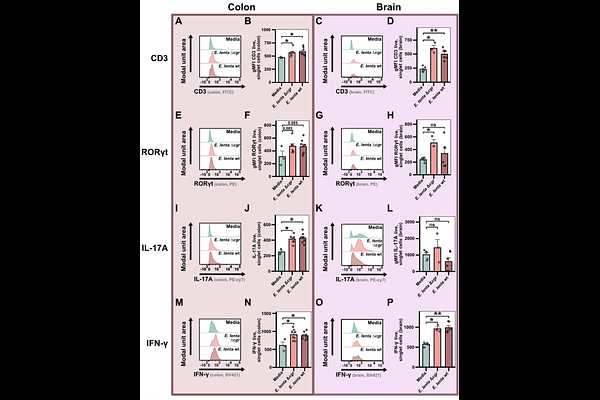
AbstractWomen are at increased risk of autoimmune diseases, including multiple sclerosis (MS); however, the degree to which sex differences in the gut microbiota impact autoimmunity remains largely unexplored. Our 27-cohort meta-analysis revealed 60 sex-associated gut bacterial species. Leveraging an independent clinical cohort, we demonstrate that female-enriched species significantly associate with MS status and clinical disability (EDSS). Top female-enriched species Eggerthella lenta drove disease in the experimental autoimmune encephalomyelitis (EAE) MS model, consistent with brain and gut lamina propria T cell infiltration and MS-associated T helper (Th) signatures. E. lenta induced intestinal Th1 and Th17 in healthy mice, independent of bacterial viability. Mechanistically, we demonstrate that TLR2 directly drives E. lenta-induced IFN-{gamma} production in Th cells and is necessary for exacerbation of EAE. Together, we identify a causal host-microbe axis contributing to sex differences in autoimmunity and provide a framework for evaluating sex as a biological variable in human microbiome research.