Disrupted glial-mediated synaptic refinement in Fragile X syndrome
Disrupted glial-mediated synaptic refinement in Fragile X syndrome
Starr, L.; Lee, M.; Vo, A.; Weisenhaus, M.; Cheadle, L. M.; Yadav, A.; Paryani, F.; Shirasu-Hiza, M.; Menon, V.; Mason, C.
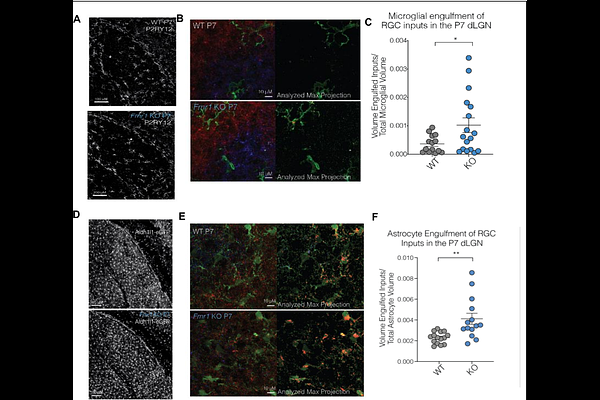
AbstractFragile X syndrome (FXS), the most common inherited cause of intellectual disability and autism, results from the loss of the RNA-binding protein fragile X mental retardation protein (FMRP). FMRP is a translational regulator and is highly expressed in glial cells, where its role in neural circuit development remains poorly defined. Here, it was observed that Fmr1 knockout mice exhibit reduced synapse size and accelerated eye-specific segregation. To examine which cell-types participate in this process, a multi-omic framework was applied to FXS model mice at postnatal day 7, a critical window for synaptic remodeling in the retinogeniculate pathway, an established model system utilized to study synaptic pruning. Single-cell transcriptomics revealed coordinated alterations in microglia, astrocytes, and neurons in genes linked to synaptic pruning. Computational modeling further demonstrated enhanced astrocyte-to-microglia signaling, particularly through Ephrin A (EphA)- and semaphorin-mediated pathways, while lipidomic profiling revealed reductions in EphA-associated lipid species required for lipid raft stability and receptor localization. Consistent with these observations, a glial engulfment assay indicated that FXS microglia and astroglia over-engulf synaptic material in the lateral geniculate nucleus, supporting the transcriptomic profile. Together, these findings identify impaired glial-driven synaptic refinement as an early mechanistic feature of FXS pathogenesis, highlighting the genes involved in this process as potential therapeutic targets during circuit development.