Development of a continuous bioreactor to maintain stable nasal microbiomes from swab specimens and synthetic communities
Development of a continuous bioreactor to maintain stable nasal microbiomes from swab specimens and synthetic communities
Ham, S.; Navarro-Diaz, M.; Camus, L.; Lucas, T. N.; Stincone, P.; Heilbronner, S.; Link, H.; Petras, D.; Huson, D.; Angenent, L. T.
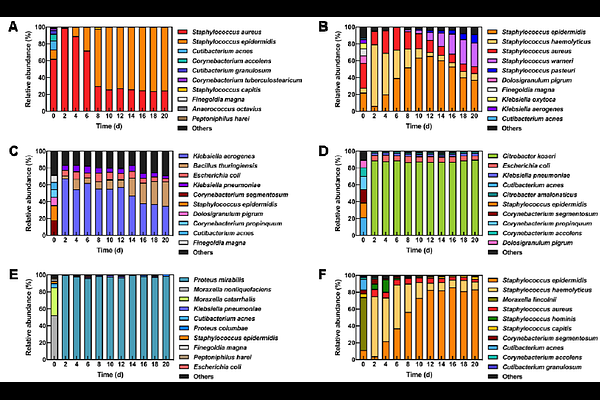
AbstractBackground: The nasal microbiome is a collection of diverse microbial populations that inhabit the nose. Staphylococcus aureus is the most common opportunistic pathogen that colonizes the nasal mucosa, increasing the risk of invasive infections in immunocompromised and hospitalized patients. Clinicians usually prescribe antibiotics to decolonize the nasal cavities of at-risk patients from S. aureus. However, their broad antimicrobial activity can damage the resident nasal microbiome. Instead, naturally occurring compounds or resident bacteria in nasal microbiomes can effectively and safely exclude S. aureus from the nose. Cell culture and animal models have been used for nasal microbiome studies. However, their unstable microbiomes reduce the accuracy and reliability of the results. Recently, continuous bioreactors have been proposed as alternatives to these models. Results: We designed and operated a continuous bioreactor system to maintain stable nasal microbiomes. Next, we inoculated the bioreactor with nasal-swab specimens that we had collected from healthy volunteers. We operated the bioreactors under varying conditions (i.e., operating mode, dilution rate, temperature, pH, and medium composition), and determined the optimal conditions (continuous mode, 1 d-1, 30, pH 6.5, and synthetic nasal medium 3), resulting in stable microbiomes consisting of the main nasal bacterial species. The nasal microbiomes in the optimized bioreactors showed high reproducibility and resilience during a pH perturbation. Moreover, all microbiomes in the bioreactor, which were inoculated with six different nasal-swab specimens, maintained stable bacterial and metabolite compositions. In addition, we applied a synthetic microbial community (SynCom), which was derived from one of the volunteers, to demonstrate a S. aureus decolonization strategy. The bioreactor, inoculated with this SynCom, maintained a stable nasal microbiome for more than one month. Finally, different S. aureus strains that we inoculated in the SynCom showed distinct growth patterns within the otherwise stable community. Conclusions: The continuous bioreactor enables the cultivation of stable nasal microbiomes for longer than one month by mimicking the environmental conditions of the human nose. The bioreactor is a valuable model for understanding the functions of the nasal microbiome and devising new decolonization strategies against S. aureus.