VE-cadherin NOT-gated CD93 CAR T cells discriminate between AML and healthy endothelial cells
VE-cadherin NOT-gated CD93 CAR T cells discriminate between AML and healthy endothelial cells
Woodring, T.; Kletzien, O. A.; Schlevensky, K.; Sanchez De Diego, C.; Vishwasrao, P.; Mahoney, L.; Kerr, S. C.; Richards, R. M.
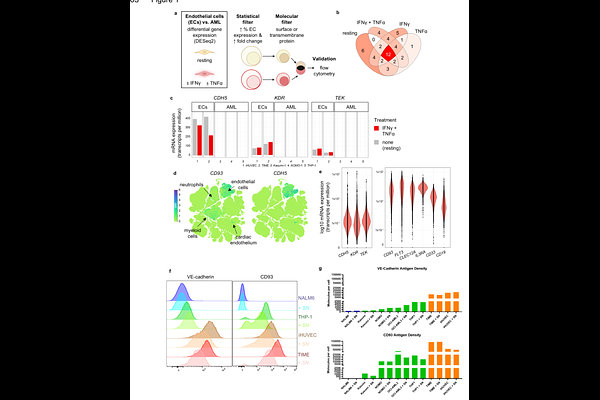
AbstractBackground: Chimeric antigen receptor (CAR) T cell therapy has transformed the treatment of B cell malignancies, but translation to acute myeloid leukemia (AML) has been hindered by on-target, off-tumor (OTOT) toxicity. In particular, endothelial cell (EC)-specific toxicity has limited clinical translation of promising leukemia stem cell-enriched targets such as CD93. Innovative strategies to mitigate EC damage while preserving antileukemic efficacy are needed. Methods: We hypothesized that a NOT-gated CAR T cell strategy could circumvent EC toxicity associated with CD93 targeting. Considering CAR target antigen density and the pro-inflammatory microenvironment of CAR T cells, we identified VE-cadherin (VC), a highly specific EC marker, as an optimal inhibitory CAR target. We engineered a novel VC-specific single chain variable fragment (scFv), confirmed EC specificity in the context of a VC-specific second-generation activating CAR, then evaluated VC/CD93 NOT-gated CAR T cells for EC protection and antileukemic activity in in vitro cytotoxicity assays and in a three-dimensional vascularized microphysiological system. Results: VC/CD93 NOT-gated CAR T cells maintain potent cytotoxicity against AML across multiple effector-to-target ratios, but preserve EC integrity, including in a three-dimensional vascular model system. Importantly, prior AML exposure did not impair the EC-protective function of the VC-specific iCAR, indicating durable NOT-gate activity under inflammatory conditions. Conversely, EC-induced iCAR inhibitory functions did not limit downstream antileukemic cytotoxicity, confirming a reversibility of both activation and inhibitory signals. Conclusions: These findings establish NOT-gated CAR T cells as an effective strategy to overcome EC-specific OTOT toxicity. Our results underscore the importance of CAR target discovery and validation across a spectrum of inflammatory states that can influence antigen expression and available therapeutic windows. This approach expands the potential CAR target landscape for AML and may be more broadly applicable to other malignancies where OTOT toxicity limits clinical translation.