Transcription termination safeguards quiescent chromatin for faithful cell-cycle re-entry
Transcription termination safeguards quiescent chromatin for faithful cell-cycle re-entry
Prashanth, A. K.; Bhardwaj, A.; Kadumuri, R. V.; Chavali, S.; Chalamcharla, V. R.
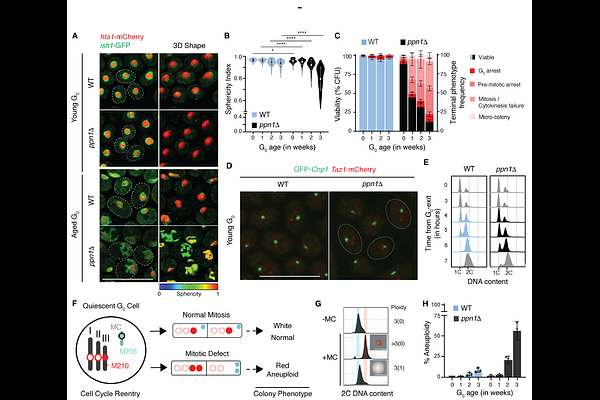
AbstractQuiescence is a conserved program of endurance and readiness in non-cycling cells that is fundamental to the longevity of eukaryotic lineages, from clonal microbial populations to human regenerative tissues. While this G0 state is universally associated with a compact chromatin organization, the active safeguards that preserve this structural template--and their necessity for future division competence and fidelity--remain unknown. Here we show, using fission yeast as a model, that chromatin structural maintenance in quiescence requires the enforcement of transcription termination by the conserved factor Ppn1PNUTS/Ref2. We identify a minimal disordered region in Ppn1 that resolves a physical conflict in the quiescent genome by preventing the transcriptional 'eviction' of cohesin. Loss of this safeguard drives a progressive structural erosion that deregulates cyclin-dependent kinase (CDK) dynamics and causes lethal aneuploidy upon cell-cycle re-entry. Notably, this deterioration is not an inevitable terminal state; re-establishing termination via a brief Ppn1 pulse resets division fidelity by stabilizing a core de novo cohesin landscape. These findings redefine quiescent chromatin as an actively maintainable blueprint rather than a passive standby state established at G0 entry. We propose that 'quiescence exhaustion' is driven by transcriptional stress eroding genomic organization, defining a structural limit to cellular longevity.