Metabolic reprogramming by caloric restriction enhances acute phase virological control and reduces chronic inflammation in SIV-infected rhesus macaques
Metabolic reprogramming by caloric restriction enhances acute phase virological control and reduces chronic inflammation in SIV-infected rhesus macaques
Suresh Babu, N.; Perdios, C.; Hallmets, M.; Brown, A. T.; Coleman, C.; Fennessey, C. M.; Allers, C.; Mostörm, M. J.; Khare, P.; Zhang, C.; Smith, B. T.; Golden, N. A.; Myers, A.; Doyle-Meyers, L.; Blaney, A.; Blair, R. V.; Saied, A. A.; Colman, R.; Keele, B. F.; Le, A.; Palmer, C. S.; Mudd, J. C.
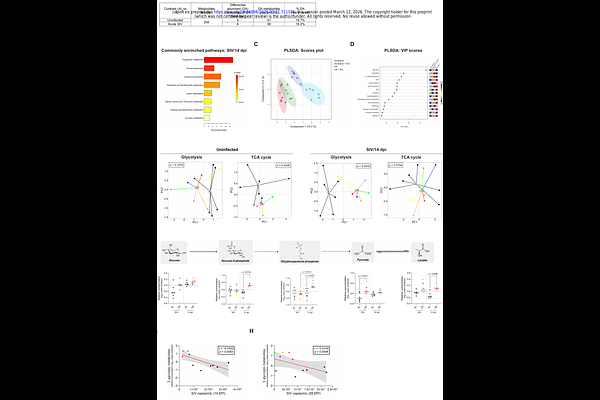
AbstractNutrient metabolism influences HIV-1 replication, antiviral immunity, and chronic inflammation, yet is difficult to leverage for therapeutic gain. We sought to modulate metabolism in the non-human primate model of HIV-1 by caloric restriction (CR), a modality canonically known for its anti-aging benefits. Four months of 30% CR was safe and resulted in broad and systemic metabolic reprogramming in healthy adult male and female rhesus macaques. Relative to that of ad libitum-fed animals, CR lowered the frequencies of target CCR5+ CD4 T cells in the gut mucosa. Upon infection with SIV, CR reduced acute-phase viremia, dampened type I interferon signaling, and overall permitted a more vigorous cycling of CD8+ T cells in lymphoid tissues. CR-induced protection from SIV was associated with a robust up-regulation of glycolysis, which supported an early reduction in viremia that ultimately waned over time. During virologic suppression with antiretroviral therapy (ART), CR significantly limited gastrointestinal (GI) immune activation, improved tricarboxylic acid cycle flux, and lowered concentrations of soluble CD14 and several TNF-related molecules in plasma. Blood SIV DNA levels, however, were unchanged by CR, suggesting that residual GI dysfunction and inflammation can be decoupled from viral persistence. Our findings highlight that a dietary modality can limit pathology in a primate lentiviral infection. They also reveal the robust but temporally constrained nature of glycolysis in supporting an acute antiviral response.