Chronic cold exposure induces plasticity of mitochondrial calcium uptake in beige and brown fat of UCP1-deficient mice.
Chronic cold exposure induces plasticity of mitochondrial calcium uptake in beige and brown fat of UCP1-deficient mice.
Chamorro, C. G.; Pathuri, S.; Acin-Perez, R.; Chhan, M.; Milner, M. G.; Ermolova, N.; Jones, A. E.; Divakaruni, A. S.; Stiles, L.; Hevener, A. L. S.; Zhou, Z.; Shirihai, O. S.; Kirichok, Y.; Bertholet, A. M.
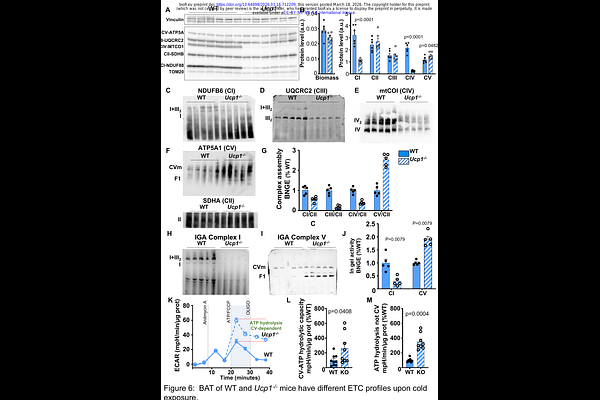
AbstractBrown adipose tissue (BAT) is a unique tissue with mitochondria specialized for thermogenesis via the BAT-specific uncoupling protein 1 (UCP1). Ucp1-/- mice cannot tolerate acute exposure to cold, illustrating the necessity of UCP1 for efficient mitochondrial thermogenesis. However, these mice adapt to low temperatures through a gradual acclimation process, suggesting a high degree of mitochondrial plasticity in brown and beige fat cells. This phenomenon, which remains to be fully elucidated, indicates the potential for these mitochondria to implement effective thermogenic mechanisms in the absence of uncoupling protein 1 (UCP1). Here, we investigated mitochondrial remodeling in beige and brown fat of Ucp1-/- mice to determine how they fulfill their thermogenic role. Upon gradual acclimation to a cold environment, Ucp1-/- mice exhibited body metabolic parameters and temperatures in the interscapular region similar to those of wild-type mice of BAT, highlighting effective thermogenesis. Interestingly, mitochondrial patch-clamp analysis and a mitochondrial Ca2+ swelling assay revealed a dramatic increase in Ca2+ uptake depending on the mitochondrial calcium uniporter (MCU) in BAT mitochondria from Ucp1-/- mice when robust thermogenesis was required. Mitochondrial remodeling was accompanied by markedly increased tethering between mitochondria and the endoplasmic reticulum (ER) in Ucp1-/- mice, confirming a significant restructuring of the contact sites between the ER and mitochondria, likely to adapt to a new Ca2+ homeostasis. Respiratory complexes also underwent significant reorganization, which partly led to a reduction in their assembly. Levels of ATP synthase and its F1 subcomplex increased, suggesting a major source of ATP consumption and energy expenditure. We propose a new role for MCU as a key regulator of mitochondrial plasticity, enabling efficient thermogenesis in beige and brown adipose tissues in the absence of UCP1.