The m6A Landscapes on the RNA of Mumps Virus and the Host 3 Modulate the Viral Replication and Antiviral Innate Immunity
The m6A Landscapes on the RNA of Mumps Virus and the Host 3 Modulate the Viral Replication and Antiviral Innate Immunity
Wang, S.; Zhang, Y.; Ping, T.; Zeng, X.; Yang, M.; Wang, W.; Hu, Q.; Fu, T.; Chen, Y.; Lu, M.
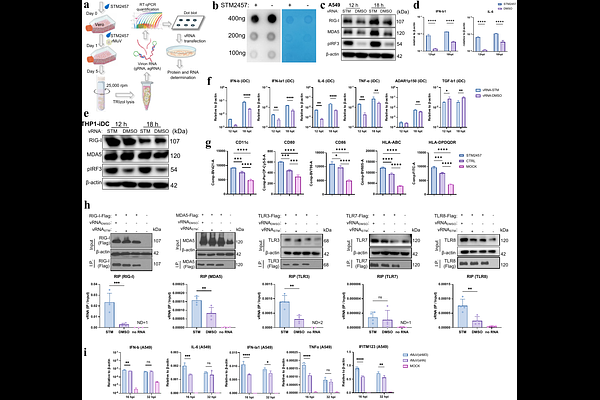
AbstractN6-methyladenosine (m6A) is the most prevalent internal modification in eukaryotic mRNA and has emerged as a critical regulator of RNA virus infection. However, its role in the mumps virus (MuV), a non-segmented negative-strand (NNS) RNA virus, remains undefined. Here, we comprehensively characterize the m6A epitranscriptome of MuV JL2 strain and its functional impact on viral replication and host innate immunity. Using single-base resolution GLORI-seq, we identified abundant m6A modifications on MuV genomic, antigenomic and messenger RNAs, with uneven distribution and non-canonical motif enrichment. Genetic depletion of METTL3 enhanced viral replication by facilitating RNA synthesis and nucleocapsid encapsidation in Vero-E6 cells. In epithelial A549 and monocytoid THP1-iDC cells, MuV infection triggered robust type I/III interferon and proinflammatory responses by activating multiple pattern recognition receptors in a m6A-dependent manner and induced incomplete iDC maturation characterized by downregulated HLA class II; however, the latter molecule can be partially restored by METTL3-knockdown. The MuV infection reshaped the host m6A landscape with a positive correlation between m6A enrichment and transcripts enhancement of numerous innate immune genes in a cell type-specific manner. Functional analyses revealed that host m6A machinery modulates antiviral signaling, which varies between viral infection and viral RNA-transfection due to the infection-induced host m6A machinery fluctuation. Collectively, our findings demonstrate that m6A serves as a bidirectional regulator during MuV infection, simultaneously constraining viral replication and modulating host immune recognition, thereby highlighting RNA methylation as a pivotal determinant of MuV pathogenesis and a potential target for optimized immunogenicity.