IFN-γ Orchestrates Coordinated Immunosuppression in Head and Neck Squamous Cell Carcinoma Through JAK-STAT-IRF8 Signaling: A Transcriptome-Wide Computational Analysis
IFN-γ Orchestrates Coordinated Immunosuppression in Head and Neck Squamous Cell Carcinoma Through JAK-STAT-IRF8 Signaling: A Transcriptome-Wide Computational Analysis
Abdelhamid, A.; Saad, e.
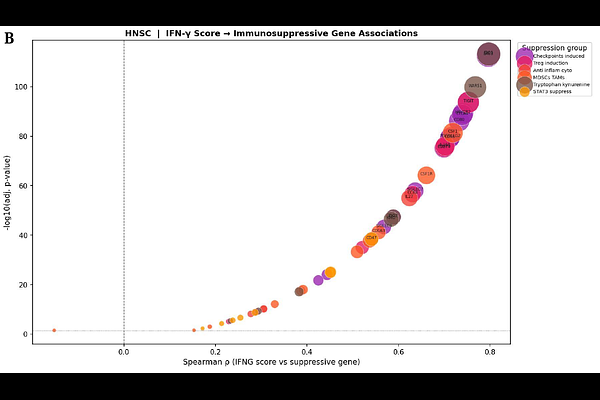
AbstractBackground: Interferon-gamma (IFN-{gamma}) is the primary effector cytokine of adaptive anti-tumor immunity, yet it paradoxically induces a potent immunosuppressive tumor microenvironment (TME). The full mechanistic scope of this paradox in head and neck squamous cell carcinoma (HNSC) has not been characterized at the transcriptomic scale. Methods: Using TCGA HNSC RNA-seq data (n = 522), we applied an integrated computational pipeline: Spearman correlation analysis, principal component analysis (PCA), UMAP, K-means clustering (k = 4), Random Forest regression, deep neural networks, permutation importance, JAK-STAT cascade mapping, and DNN-based transcriptome-wide mediation analysis across 57 IFN-{gamma} pathway and 78 immunosuppressive genes. Results: IFN-{gamma} pathway activity was universally and positively correlated with six immunosuppressive axes, including checkpoints (CD274; LAG3; IDO1), Tregs, myeloid suppression, and tryptophan catabolism. K-means clustering identified four immunologically distinct tumor subgroups. DNN models predicted suppressive TME. Permutation importance identified IRF8 as the dominant mediator linking IFN-{gamma} signaling to immunosuppression. DNN mediation analysis identified PDCD1LG2 (PD-L2) as the strongest intermediary between IFNG and PD-L1 regulation, followed by JAK2 and GBP5. Conclusions: IFN-{gamma} orchestrates coordinated immunosuppression in HNSC through JAK-STAT-IRF8 signaling. PDCD1LG2 and JAK2 are actionable mediators of this paradox, supporting combination strategies co-targeting IFN-{gamma}-induced checkpoint induction and direct checkpoint blockade in HNSC immunotherapy.