Photopatterned Sacrificial Vascular Architectures for Large Tissue-Scale Oxygenation
Photopatterned Sacrificial Vascular Architectures for Large Tissue-Scale Oxygenation
Coates, I. A.; Kohnke, C. A.; Tan, Y. L.; Alnasir, D. I.; Nguyen, A. N.; Heng, E. E.; Kwan, A.; Dualy, M. T.; Schaar, B.; Skylar-Scott, M. A.; MacArthur, J. W.; Shaqfeh, E. S. G.; DeSimone, J. M.
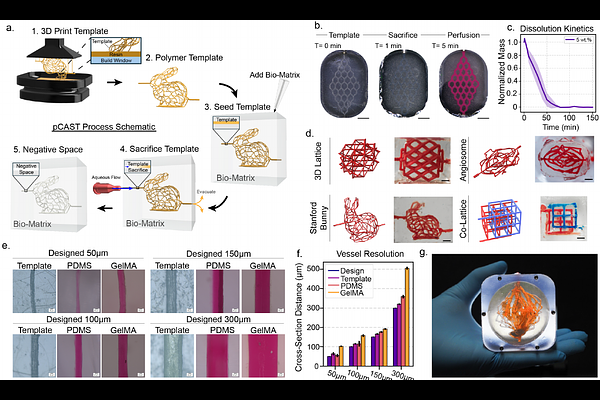
AbstractThe engineering of thick, metabolically active tissues is constrained by the lack of scalable methods to create perfusable vasculature. This hinders effective metabolite transport in large tissue volumes, posing a critical barrier for regenerative tissue applications. In this study, we introduce photopatterned Channel Architectures with Sacrificial Templates (pCAST), an additive manufacturing strategy for generating three dimensional (3D), interconnected vascular networks with precisely defined negative space. Water-soluble sacrificial templates were fabricated using scalable Continuous Liquid Interface Production (CLIP), embedded within tissue constructs, and flushed away to yield 50 um perfusable channels spanning centimeter-scale tissue constructs. We then apply experimental oxygen mapping and viability analysis to pCAST constructs to build finite-element models that predict patterns of oxygen availability and tissue survival are governed by the balance between metabolic demand and vascular architecture, consistent with reaction-diffusion theory. This computational framework quantitatively predicts oxygen distributions and viability boundaries across vascular geometries and is validated experimentally. Together, these results establish pCAST as a scalable design framework linking vascular architecture, perfusion, and metabolic support for engineering large, 3D perfused tissue constructs.