A comprehensive CRISPR screen of the Drosophila glutamate receptome reveals Ekar as a selective regulator of presynaptic homeostatic plasticity
A comprehensive CRISPR screen of the Drosophila glutamate receptome reveals Ekar as a selective regulator of presynaptic homeostatic plasticity
Martinez, J.; Chien, C.; Dong, W.; Tran, N.; Chang, A.; Zak, H.; Shaw, S.; Shohat-Ophir, G.; Dickman, D.
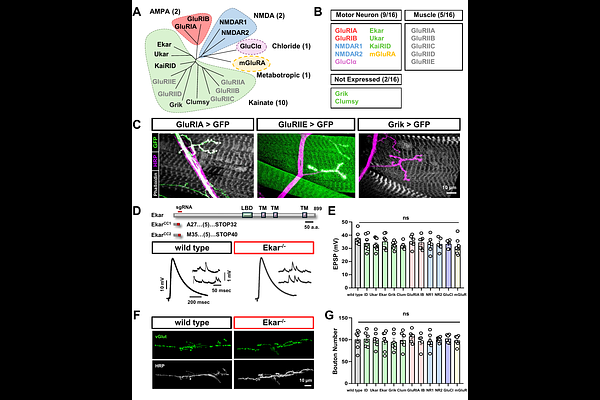
AbstractHomeostatic mechanisms protect synapses from destabilizing challenges throughout an organism's lifespan, ensuring stable yet flexible neural network activity. To delineate the molecular basis of presynaptic homeostatic potentiation (PHP), we conducted a comprehensive, in vivo CRISPR/Cas9-based screen of all 16 glutamate receptor (GluR) genes encoded in the Drosophila genome. We first generated a complete expression atlas across larval and adult stages, identifying nine GluRs expressed in presynaptic motor neurons. We then generated null mutants for all 16 GluRs and screened them at the larval neuromuscular junction. While the loss of any single presynaptic GluR did not affect baseline synaptic growth or neurotransmission, our screen revealed a selective and critical requirement for the kainate receptor subunit ekar in the expression of chronic PHP. Further genetic analysis indicates that Ekar functions coordinately with the kainate receptor subunits KaiRID and Ukar within a shared pathway to promote this plasticity. Mechanistically, Ekar acts downstream of active zone remodeling to drive the homeostatic enhancement of presynaptic Ca2+ influx, which is the defining feature of chronic PHP. Together, this genome-wide analysis establishes a definitive functional atlas for the Drosophila glutamate receptome and highlights a specialized, essential role for Ekar in stabilizing long-term synaptic homeostasis.