A structure-guided pipeline yields peptide inhibitors that disarm fungal peptidase-driven virulence and resistance
A structure-guided pipeline yields peptide inhibitors that disarm fungal peptidase-driven virulence and resistance
Gutierrez-Gongora, D.; Hambly, J.; Chan, N.; Romero, O.; Woods, M.; Olabisi-Adeniyi, E.; Dawe, A.; Mantilla, M. J.; Wasney, G. A.; Deyarmin, J.; Samra, S. N.; Valiente, P. A.; Alpaugh, M.; Sellam, A.; Prosser, R.; Hamilton-Wright, A.; Geddes-McAlister, J.
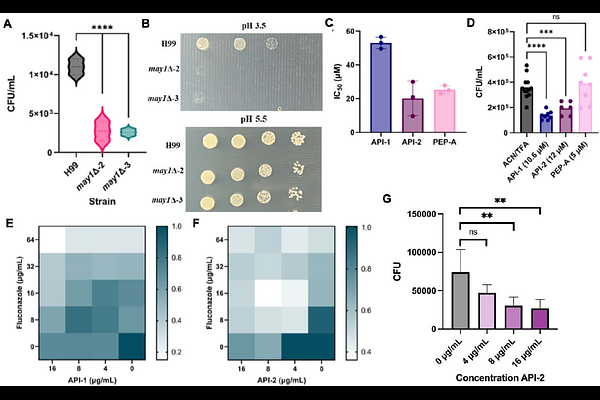
AbstractFungal infections are a major global health challenge, with current antifungal therapies limited by toxicity, cost, and resistance. For Cryptococcus neoformans, key virulence factors that initiate and sustain infection are regulated by fungal peptidases to produce a polysaccharide capsule, promote immune evasion, and support antifungal resistance. These peptidases represent promising targets for antivirulent therapeutic strategies. Here, we developed a computational pipeline to predict and design peptide- and protein-based inhibitors against cryptococcal peptidases. Specifically, we targeted three virulence-associated peptidases: Rim13 (cysteine), May1 (aspartic), and CnMpr1 (metallo). Cysteine peptidase inhibition decreased capsule/cell size ratios without impeding fungal growth and reduced fungal survival within macrophages. Similarly, aspartic peptidase inhibition enhanced fungal clearance within alveolar macrophages and disrupted biofilm formation with additive effects towards fluconazole susceptibility in resistant strains. Additionally, metallopeptidase inhibition through catalytic zinc chelation and blocked substrate binding led to enhanced enzymatic inhibition and reduced in vitro blood-brain barrier crossing. Moreover, an in vivo larval model assessing inhibitor efficacy produced additive effects with fluconazole and lacked host cell cytotoxicity and fungicidal properties, reinforcing anti-virulence mechanisms and therapeutic potential while limiting the evolution of resistance. Further, global proteome profiling of inhibitor treated cells defined a mechanism of cell wall disruption, impeding fungal virulence. Taken together, the designed peptidase inhibitors exhibited potent antifungal activity without harming mammalian cells, establishing a predictive framework for rational scaffold design of next-generation antifungals that disarm the pathogen enabling immune-mediated clearance.