Likely role of promoter reconstitution in Mpr-mediated D29 resistance by Mycobacterium smegmatis
Likely role of promoter reconstitution in Mpr-mediated D29 resistance by Mycobacterium smegmatis
Yusuf, B.; Ju, Y.; zhou, B.; Malik, A.; Alam, M. S.; Li, L.; Abraha, H. T.; BELACHEW, A. M.; Fang, C.; Tian, X.; Chen, H.; Wan, L.; Feng, L.; Xiong, X.; Wang, S.; Zhang, T.
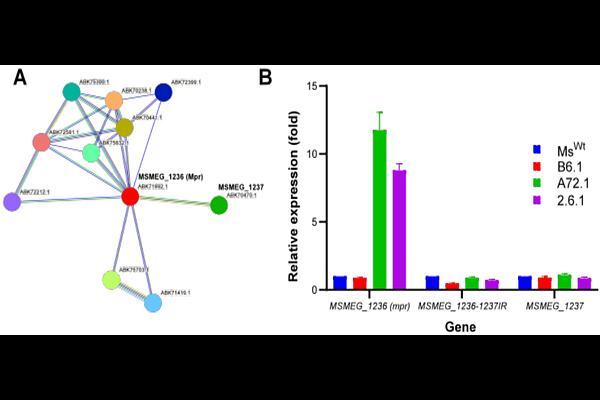
AbstractThe multi-copy phage resistance gene (mpr) of M. smegmatis is a major factor in resistance to the lytic mycobacteriophage D29. Mpr is a membrane-bound exonuclease that cleaves phage DNA post injection, hence blocking downstream stages in the phage infection cycle. The mechanism of resistance allows for adsorption, is non-abortive and independent of any mutation in the gene. Rather, it depends on overexpression of a wild type copy of the gene. However, the underlying factor behind mpr overexpression in spontaneous D29-resistant mutants of M. smegmatis remained elusive. Here, we report that D29 infection triggers insertion sequence (IS) rearrangements, including the transposition and integration of IS6120 directly upstream of mpr. Mutants with IS6120 integration upstream of mpr show highly elevated Mpr expression. Whole genome sequence analysis reveals that IS6120 introduced a putative transcription factor-binding site and a canonical -35 promoter element at the integration site, hence reconstituting a fuller promoter (rcp) than the original promoter (wtp) at the site. Promoter reporter assays suggest that rcp is a far stronger promoter than wtp, implying that elevated mpr expression in D29-resistant mutants with this transposition event could be due to promoter reconstitution. While strains with this transposition event appear to grow normally, rcp-driven, vector-borne Mpr overexpression appears to be toxic as it barely allows for colony formation on agar plates. This study reports a previously unknown factor likely behind mpr regulation in M. smegmatis, adding to the existing knowledge of mycobacterial anti-phage defense mechanisms and guiding rational phage engineering efforts for therapeutic applications.