Single-cell full-length transcriptome of human lung reveals genetic effects on isoform regulation beyond gene-level expression
Single-cell full-length transcriptome of human lung reveals genetic effects on isoform regulation beyond gene-level expression
Li, B.; Luong, T.; Sisay, E.; Yin, J.; Zhang, Z. E.; Vaziripour, M.; Shin, J. H.; Zhao, Y.; Tran, B.; Byun, J.; Li, Y.; Lee, C. H.; O'Neill, M.; Andresson, T.; Chang, Y. S.; Gazal, S.; Landi, M. T.; Rothman, N.; Long, E.; Lan, Q.; Amos, C. I.; Zhou, A. X.; Zhang, T.; Lee, J. G.; Shi, J.; Mancuso, N.; Xia, J.; Zhang, H.; Kim, E. Y.; Choi, J.
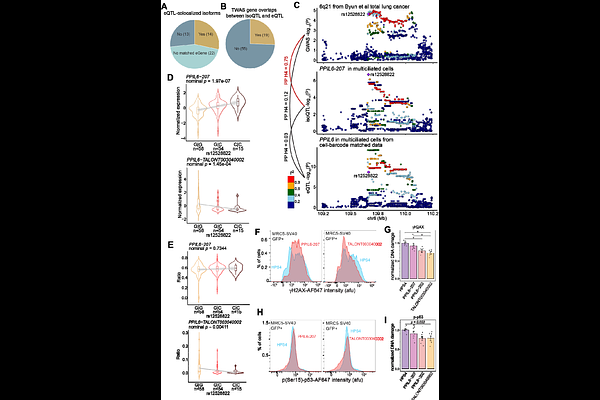
AbstractGenetic regulation of splicing uniquely contributes to trait-associated genome-wide association studies (GWAS) signals. However, quantitative trait loci (QTL) analysis using short-read sequencing of bulk tissues fails to capture full-length and cell-type-specific isoforms. Here, we present an isoform-level lung cell atlas from 129 never-smoking Korean women using single-cell long-read RNA-sequencing, identifying abundant unannotated and cell-type-specific isoforms. Isoform-level signatures of 37 lung cell types display a larger difference and therefore improve cell-type classification compared to gene-level expression. Notably, isoform-QTLs (isoQTLs) detect unannotated and/or cell-type-specific isoforms with independent genetic regulation from expression-QTL (eQTL), supported by enriched splicing functional elements. IsoQTLs nominate susceptibility isoforms from previously unexplained lung function and cancer GWAS loci, via eQTL-independent signals. We highlight a potentially functional novel variant of PPIL6 in multiciliated cells underlying lung cancer risk through alternative splicing. This isoform-level resource advances our understanding of cell-type-specific isoform regulation and its contribution to lung traits and diseases.