Volumetric mechanosensing of CAF in 3D hydrogels drive altered drug response in breast cancer
Volumetric mechanosensing of CAF in 3D hydrogels drive altered drug response in breast cancer
Devarasou, S.; Sung, N. J.; Ham, S. H.; Kiwanuka, M.; Young, J. L.; Shin, J. H.
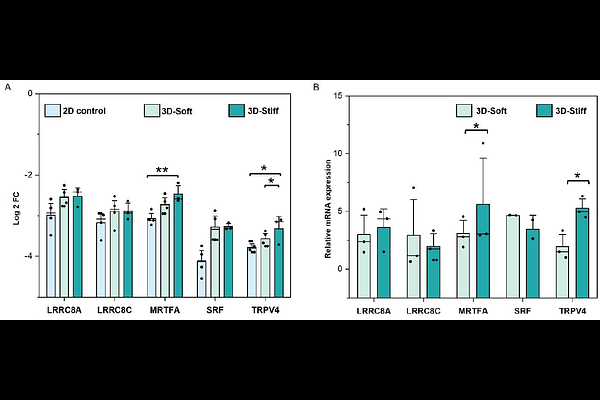
AbstractAltered mechanical properties of the tumor microenvironment (TME) influence cancer progression, yet the mechanistic basis by which 3D mechanics shape CAF heterogeneity and downstream tumor drug response remains poorly understood. Here, we engineered a modulus-tunable gelatin methacryloyl (GelMA) hydrogel platform spanning a normal-like (soft ~2 kPa) to desmoplastic-like (stiff~40 kPa) range to culture primary breast CAFs under 3D confinement. CAFs exhibited pronounced volumetric morphoadaptation across matrices, with soft 3D matrices supporting larger, more protrusive morphologies and stiff gels constraining cell geometry. In contrast to canonical 2D paradigms, nuclear YAP localization was reduced in stiff 3D matrices and varied substantially across cells, consistent with a dominant role for 3D geometric/volumetric state in regulating mechanotransduction. Functionally, in transwell co-culture with MCF-7 spheroids under paclitaxel treatment, CAFs cultured in stiff 3D matrices induced a broader chemoresistance-associated transcriptional program, whereas soft 3D matrices CAFs favored stress/checkpoint-like responses. A 2D monolayer comparator indicated that coordinated resistance-associated programs emerge most clearly in 3D tumor architecture. Together, these results establish a GelMA-based biomaterials framework in which CAF volumetric state provides a quantifiable intermediate linking 3D matrix mechanics to mechanotransduction and tumor drug-response programs, motivating future strategies to modulate stromal function through mechanically controlled cell-state regulation.