Alternative splicing of a TPR domain determines mitochondrial versus plastid function of the only CLU family protein in Marchantia polymorpha
Alternative splicing of a TPR domain determines mitochondrial versus plastid function of the only CLU family protein in Marchantia polymorpha
Lozano-Quiles, M.; Raval, P. K.; Gould, S. B.
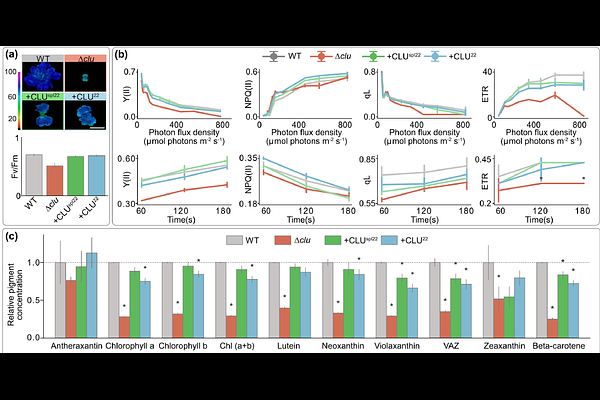
AbstractIn plant cells, the multi-domain proteins FRIENDLY and REC regulate the cellular organization, distribution and proliferation of mitochondria and plastids, respectively. Both proteins share a similar overall domain architecture and belong to the larger CLUSTERED MITOCHONDRIA (CLU) superfamily. Domains of CLU proteins have been shown to interact with translation related proteins, tRNA synthetases and even mRNA, but their exact modes of operation remain cryptic and how organelle specificity of CLU paralogs in plant cells is achieved unknown. We characterized the single CLU family protein of the liverwort Marchantia polymorpha that we demonstrate to be transcribed either with or without exon 22, which changes the configuration of the TPR domains in the C-terminus. Knockout of MpCLU affects both mitochondria and plastids, and independent rescues show that the splice variant with exon 22 (MpCLU22) serves mitochondrial- and the one lacking exon 22 (MpCLUspl22) plastid biology. The CLU-C domain of the protein is responsible for nuclear localisation and expressed alone induces a phenotype that differs in photosynthesis performance and transcriptome changes from that of the knockout of MpCLU. Our results identify the C-terminal TPR motif to be responsible for organelle specificity in plants and they provide an example of how genome reformatting and gene loss can be compensated for by the alternative splicing of a single exon.