Bromodomain protein IBD1 bridges histone acetylation and H2A.Z deposition to fine-tune transcription and prevent hyperactivation in Tetrahymena thermophila
Bromodomain protein IBD1 bridges histone acetylation and H2A.Z deposition to fine-tune transcription and prevent hyperactivation in Tetrahymena thermophila
Zhang, Z.; Li, H.; Wei, F.; Ju, A.; Jiang, H.; Ye, F.; Liu, Y.; Gao, S.; Wang, Y.
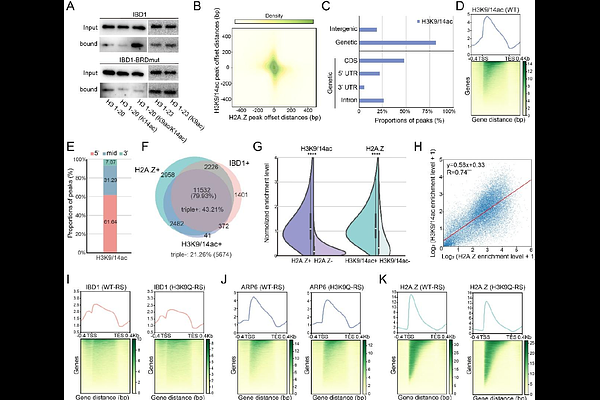
AbstractEukaryotic gene expression is dynamically regulated through the interplay between histone modifications and chromatin remodeling, yet how these processes are coordinated remains incompletely understood. Here, we identify IBD1 as a critical adaptor that bridges histone acetylation and SWR-mediated H2A.Z deposition. We demonstrate that the bromodomain of IBD1 recognizes histone acetylation, specifically H3K9/14 di-acetylation to recruit the SWR complex subunit ARP6, ensuring precise H2A.Z incorporation into chromatin. Genetic disruption of IBD1, either by deletion or bromodomain mutation, attenuates H2A.Z occupancy at target loci, confirming its essential role in H2A.Z deposition. Strikingly, perturbation of the histone acetylation-IBD1-H2A.Z axis triggers transcriptional hyperactivation, revealing a dual function of H2A.Z in sustaining basal transcription while preventing aberrant gene induction. This defines a novel regulatory paradigm in which IBD1 couples acetyl-mark decoding to SWR-dependent H2A.Z deposition, establishing transcriptional homeostasis. Our findings resolve a central ambiguity by demonstrating that H2A.Z acts as a repressive buffer in highly acetylated regions, counterbalancing the inherent activation potential of acetylation. This study provides a framework for investigating H2A.Z functions and reveals new perspectives on how chromatin modifications and histone variants interplay in transcriptional regulation.