An N-degron proteolytic pathway modulates recipient susceptibility to T6SS DNase effectors
An N-degron proteolytic pathway modulates recipient susceptibility to T6SS DNase effectors
Wen, Y.-H. V.; Lin, H.-H.; Zheng, X.-T.; Hwang, H.-H.; Lai, E.-M.
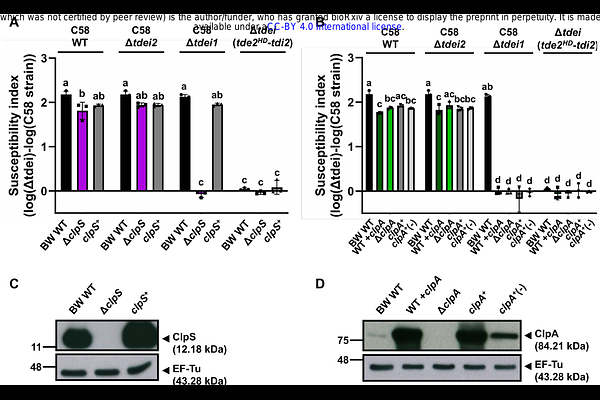
AbstractThe type VI secretion system (T6SS) is a contractile nanoweapon widely employed by Gram-negative bacteria to gain competitive advantages by injecting effector proteins into recipient cells. Although the biochemical activities of T6SS effectors have been well characterized, how recipient factors modulate effector toxicity remains poorly understood. Using Agrobacterium C58 as a model, previous work identified the Escherichia coli ClpAP protease as a recipient susceptibility (RS) factor that enhances T6SS-mediated interbacterial competition. Agrobacterium C58 deploys two DNase effectors, Tde1 and Tde2, as the major antibacterial weapon. Here, we demonstrate that the recipient ClpAP protease and its adaptor ClpS enhanced C58-mediated interbacterial competition in a Tde2-dependent manner in both intra- and interspecies competition. Ectopic expression of Tde2 in E. coli caused growth inhibition and DNA cleavage in the presence of a functional ClpAPS protease complex, but not in any of the clpP, clpA or clpS mutants. Notably, Tde2 accumulated in these mutants but not in wild-type cells, whereas a catalytic variant accumulated regardless of ClpAPS status, suggesting that Tde2 is not directly degraded by ClpAPS. Instead, Tde2 depends on ClpAPS for full toxicity, likely through degradation of inhibitory N-degron substrate(s). Affinity purification of His-tagged Tde2 in a clpP mutant background, followed by mass spectrometry, identified eight N-degron substrate candidates. Tde2-mediated interbacterial competition was significantly reduced by overexpression of three candidates. Among them, the Tde2 DNase domain directly associated with guanosine 5'-monophosphate reductase GuaC, supporting a model in which Tde2 toxicity is blocked by binding of GuaC. Collectively, our findings reveal an unanticipated layer of recipient-mediated regulation in T6SS competition and highlight proteolytic control of inhibitory substrates as a determinant of bacterial susceptibility during interbacterial conflict.