A novel 3D-printed hydrogel platform for controlled delivery of BMP-9 coated calcium sulfate microparticles with co-delivery of preosteoblasts from a cell encapsulated coating layer
A novel 3D-printed hydrogel platform for controlled delivery of BMP-9 coated calcium sulfate microparticles with co-delivery of preosteoblasts from a cell encapsulated coating layer
Unagolla, J. M.; Jayasuriya, A. C.
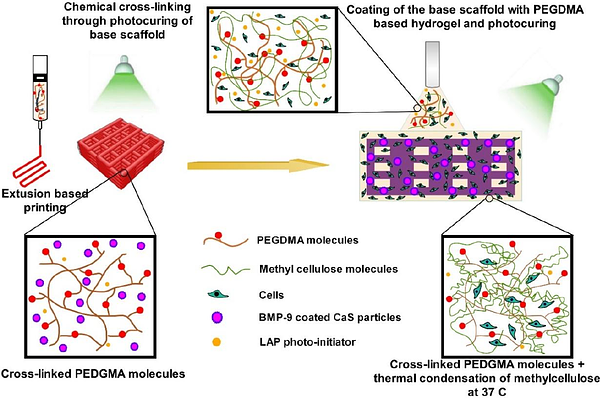
AbstractAbstract: Controlled delivery of growth factors and viable cells remains a significant challenge in bone tissue engineering. In this study, a 3D-printed hydrogel scaffold system was developed for the co-delivery of bone morphogenetic protein-9 (BMP-9) and preosteoblasts to enhance bone regeneration. The system consisted of a 3D-printed base scaffold containing BMP-9-coated calcium sulfate (CaS) microparticles and a photocurable hydrogel coating layer encapsulating viable cells. The scaffold design exploited electrostatic interactions between BMP-9 and gelatin matrices by incorporating gelatin type B in the base scaffold and gelatin type A in the coating layer. Differences in the isoelectric points of these gelatin types were utilized to regulate protein binding and release. Release studies demonstrated that CaS microparticles alone exhibited rapid burst release, with nearly 80% of BMP-9 released within 24 h. Encapsulation of BMP-9 coated CaS particles in the 3D-printed scaffolds reduced the release rate, while the addition of the coating layer significantly improved sustained release, limiting BMP-9 release to approximately 50-60% by day 5. Bioactivity studies showed enhanced cell attachment in BMP-9 containing scaffolds compared with controls. Live/Dead cytotoxicity assays demonstrated high cell viability (>80%) within the coating layer over the culture period, confirming that the encapsulation and photocuring processes did not adversely affect cell survival. Cell proliferation and differentiation were further evaluated using WST-1 and alkaline phosphatase assays. The results demonstrate that electrostatic interactions governed by gelatin type selection can regulate BMP-9 release while maintaining high cell viability, providing a promising platform for growth factors and cell delivery in bone tissue engineering.