Inhibition of p107 alleviates liver steatosis by reducing de novo fatty acid synthesis.
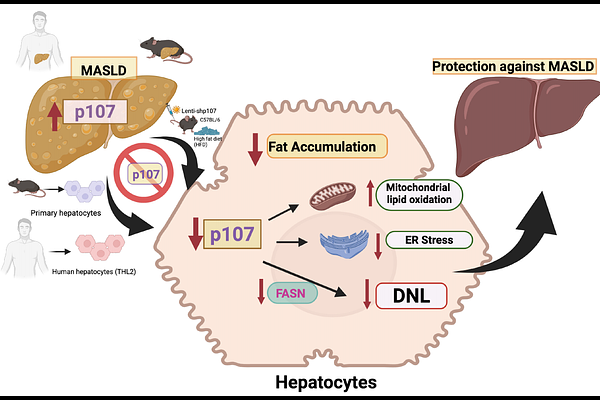
Inhibition of p107 alleviates liver steatosis by reducing de novo fatty acid synthesis.
Cunarro, J.; Miguens, M. V.; de Oliveira-Diz, T.; Buque, X.; Oro, L.; Riobello, C.; Iglesias Moure, J.; Quintela-Vilarino, C.; Maduro, A. T.; Cabaleiro, A.; Novoa, E.; Fuentes-Iglesias, A.; Fidalgo, M.; Guallar, D.; Vidal, A.; Mora, A.; Varela-Rey, M.; Nogueiras, R.; Sabio, G.; Aspichueta, P.; Fajas, L.; Dieguez, C.; Tovar, S.
Abstractp107 expression is increased in patients with MASLD and correlates with disease severity. In mouse models, global and liver-specific p107 deficiency protect against high-fat diet-induced steatosis without affecting body weight. This is associated with reduced expression of lipogenic enzymes including fatty acid synthase (FASN), and enhanced mitochondrial oxidative pathways. Conversely, hepatic restoration of p107 reversed these effects and promoted lipid accumulation and endoplasmic reticulum stress. Consistent with this in human hepatocytes, p107 silencing reduces lipid accumulation, decreases DNL and enhances mitochondrial respiration, whereas p107 overexpression induces the opposite phenotype. Notably, FASN knockdown attenuates the pro-steatotic effects of p107, indicating that it is a critical downstream mediator of p107. Together, these findings establish p107 as a physiological regulator of hepatic lipid metabolism, with its dysregulation contributing to the development of MASLD p107 expression is increased in patients with MASLD and correlates with disease severity. In mouse models, global and liver-specific p107 deficiency protect against high-fat diet-induced steatosis without affecting body weight. This is associated with reduced expression of lipogenic enzymes including fatty acid synthase (FASN), and enhanced mitochondrial oxidative pathways. Conversely, hepatic restoration of p107 reversed these effects and promoted lipid accumulation and endoplasmic reticulum stress. Consistent with this in human hepatocytes, p107 silencing reduces lipid accumulation, decreases DNL and enhances mitochondrial respiration, whereas p107 overexpression induces the opposite phenotype. Notably, FASN knockdown attenuates the pro-steatotic effects of p107, indicating that it is a critical downstream mediator of p107. Together, these findings establish p107 as a physiological regulator of hepatic lipid metabolism, with its dysregulation contributing to the development of MASLD.