Immune reprogramming of 3D tumor models via optoporation-mediatedtargeted gene delivery to macrophages
Immune reprogramming of 3D tumor models via optoporation-mediatedtargeted gene delivery to macrophages
Poljak, I.; Hussein, I. N.; Gu, C.; Giustarini, G.; Teng, X.; Toyama, Y.; Chiappini, C.; Adriani, G.
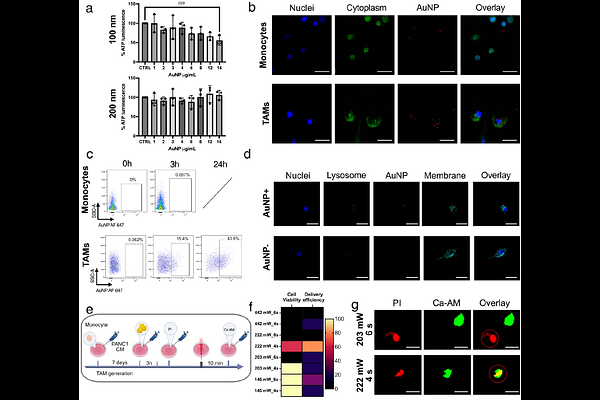
AbstractThe dynamics of the tumor microenvironment (TME) are a key determinant of cancer progres-sion and therapeutic resistance through complex interactions between tumor, stromal and im-mune cell populations. Among these, tumor-associated macrophages (TAMs) play a central role in promoting tumor growth and immune suppression. However, the specific contributions of TAMs remain poorly understood due to the lack of tools enabling selective genetic manipulation in three-dimensional (3D) tumor models. Here, we present a gold nanoparticle-assisted opto-poration approach that enables spatially selective plasmid-based gene delivery to TAMs within intact heterocellular 3D pancreatic ductal adenocarcinoma (PDAC) spheroids, thereby modulat-ing the TME. In two-dimensional (2D) TAM cultures, conventional transfection of IRF5- and IKBKB-encoding plasmids validated their capacity to induce TAM repolarization, as evidenced by activation of interferon signaling. Extending this approach to 3D PDAC spheroids, nanopar-ticle-assisted optoporation achieved selective transfection of TAMs with IRF5- and IKBKB-encoding plasmids by transiently generating nanoscale membrane pores in illuminated cells. TAMs transfection elicited a robust interferon response, marked by transcriptional upregulation of IFNA, IFNB1, and CXCL10, and increased protein levels of IFNB1, IFNL1, and CXCL13, together with downregulation of pro-tumorigenic markers CEACAM5, IL19, and IL32. These coordinated changes indicate a shift towards an anti-tumorigenic TME. By enabling minimally invasive, TAM-specific gene delivery in complex multicellular 3D spheroids, this strategy al-lows precise modulation of the TME and opens new avenues for modeling its dynamics in can-cer progression and therapeutic response.