Neuronal ketone body utilization couples exercise and time-restricted feeding to cognitive enhancement
Neuronal ketone body utilization couples exercise and time-restricted feeding to cognitive enhancement
Salathe, S. F.; Kugler, B. A.; Franczak, E.; Davis, X. C.; Boakye, F. B.; Allen, J.; Fulghum, K. L.; Queathem, E. D.; Morris, E. M.; Puchalska, P.; Crawford, P. A.; Thyfault, J.
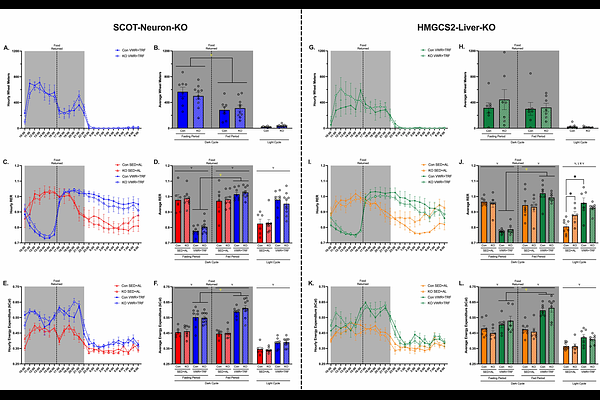
AbstractKetogenesis and ketone body metabolism are linked to brain health benefits, including delaying age-related cognitive decline and neurodegeneration. Exercise, particularly when combined with an overnight fast, stimulates ketogenesis and ketone body turnover as well as improves brain metabolism and cognition. Yet, whether ketone metabolism is obligatory for this response is unknown. Here, we use chronic exercise via voluntary wheel running plus time-restricted feeding (VWR+TRF, fasting from ZT10.5-18.5) to explore whether ketone bodies are a potential mediator of exercise-induced brain health benefits in middle-aged mice. To independently distinguish the roles of neuronal ketone body metabolism vs. hepatic ketone body production, we studied middle-age female neuronal-specific SCOT knockout mice and female hepatocyte-specific HMGCS2 knockout mice, respectively. VWR+TRF was compared to sedentary ad-libitum fed (SED+AL) mice to assess the impact on whole-body metabolism (indirect calorimetry), cognition (Barnes Maze and Y-Maze), and molecular adaptations in the hippocampus (proteomics). VWR+TRF robustly upregulated systemic lipid oxidation in all mice, regardless of genotype, during the first 6.5 hours of the dark period. In female SCOT-Neuron-KO mice, we show impaired responses to VWR+TRF in indices of short- and long-term memory. Proteomic analysis of isolated hippocampi revealed that SCOT-Neuron-KO mice failed to globally upregulate key facilitators of synaptic function, including leucine-rich repeated transmembrane proteins, neurexins, and neuroligins. In female HMGCS2-Liver-KO mice, impaired responses to VWR+TRF in indices of short-term memory were paired with an upregulation in ketogenesis machinery in the hippocampal proteome, suggesting potential in vivo evidence of cerebral ketogenesis, a mechanism mitigating an otherwise more pronounced behavioral phenotype. Together, these findings suggest that neuronal ketone body utilization is essential, and hepatic ketone production is contributory, to the full cognitive and synaptic adaptations to exercise plus time-restricted feeding, supporting ketone metabolism as a key mechanistic link between metabolic state and brain health in midlife.