Shifting Perspectives on Biotherapeutic Treatment in Ulcerative Colitis using Lipid Mesophases: Formulation Design and Preclinical Validation
Shifting Perspectives on Biotherapeutic Treatment in Ulcerative Colitis using Lipid Mesophases: Formulation Design and Preclinical Validation
Rafaela, G.; Tiziana, C.; Reto, C.; Emily, C.; Marlene, S.; Rogler, G.; Scharl, M.; Bergadano, A.; Mezzenga, R.; Kuentz, M.; Luciani, P.; Aleandri, S.
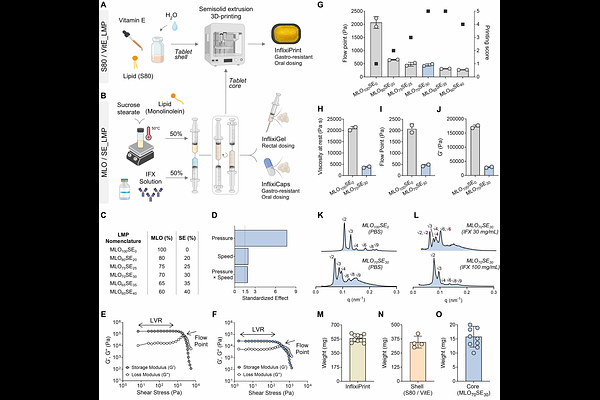
AbstractBiotherapeutics are required to achieve high remission rates in patients with severe ulcerative colitis (UC); however, adverse effects, complex dosing regimens, administration routes, and low patient compliance may limit their widespread clinical use. Given the localized nature of UC, this study aimed to develop and evaluate a localized delivery strategy for infliximab (IFX), an anti tumor necrosis factor alfa (TNF alpha) monoclonal antibody (mAb) recommended for moderately-to-severely active UC. Exploiting the intrinsic biocompatibility, mucoadhesivity, and protein-entrapment capacity of lipid mesophases (LMPs), IFX was encapsulated within the gel matrix, providing protection against enzymatic and environmental degradation. IFX-loaded LMPs were designed for targeted delivery to inflamed colonic tissues via rectal or oral administration, with patient-centric oral dosage forms manufactured using a 3D printing approach. A comprehensive physicochemical characterization was performed to elucidate mesophase self-assembly and its relationship with IFX release profiles in biorelevant fluids. Therapeutic efficacy was evaluated in vivo using a dextran sulphate sodium (DSS) induced colitis rat model, which demonstrated rectal gel retention for at least 8 h and colonic targeting of the oral formulation within 6 h. Under severe inflammatory conditions, LMP based formulations reduced disease activity, inflammatory biomarkers (TNF alpha and fecal lactoferrin), and colon shortening to values comparable to those of healthy controls, outperforming the therapeutic efficacy of subcutaneous IFX. Overall, this study establishes a biocompatible delivery platform that enables targeted colonic IFX release and suppresses systemic absorption, representing a promising advancement in the biotherapeutic treatment of UC.