Modes of programmed macrophage cell death govern outcome of cutaneous wound healing
Modes of programmed macrophage cell death govern outcome of cutaneous wound healing
Injarabian, L.; Reiche, N.; Willenborg, S.; Welcker, D.; Bai, Y.; Schoenenberg, E.; Sanin, D. E.; Tanevski, J.; Pasparakis, M.; Kashkar, H.; Eming, S. A.
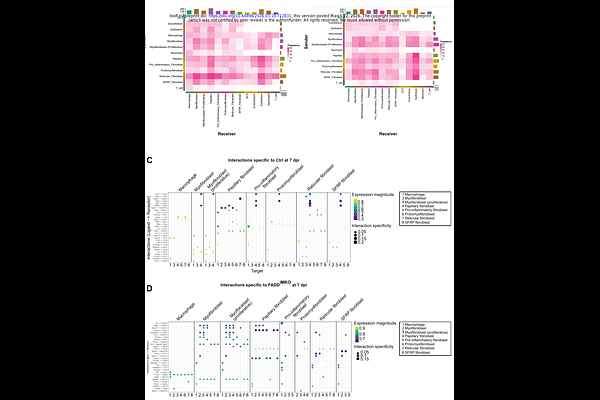
AbstractMisregulation of tissue repair programs can severely compromise repair outcome. Timely clearance of inflammatory macrophages through regulated cell death is a prerequisite for resolution of inflammation and successful repair. How different modes of macrophage regulated cell death regulate repair and direct healing outcome remains unclear. Using inducible genetic models to trigger macrophage necroptosis (FADDiMKO) or enhance apoptosis of macrophages (cIAP1iMKOcIAP2-/-), we reveal opposing effects on the architecture of the wound tissue. Macrophage necroptosis profoundly disrupted tissue organization: FADDiMKO wounds exhibited reduced numbers of reparative (IL-4RLy6Clow) macrophages, failed inflammatory resolution, accompanied by a hemorrhagic granulation tissue and reduced myofibroblast differentiation in the mid-phase of repair. In striking contrast, excessive apoptosis preserved fundamental tissue architecture and vascular integrity, and reduced scar formation. Furthermore, single-cell transcriptomics demonstrated that macrophage necroptosis disrupts tissue-wide cellular communication networks essential for coordinated repair, in particular macrophage-fibroblast crosstalk. These findings establish that wound healing quality depends on both mode and rate of macrophage programmed death, providing a framework for therapeutic targeting of macrophages in wound healing disorders and fibrotic diseases.