Unraveling Tissue-Specific Molecular Signatures and Convergent Pathway Enrichments in Suicidal Behavior
Unraveling Tissue-Specific Molecular Signatures and Convergent Pathway Enrichments in Suicidal Behavior
Jenkins, A. K.; Jia-Richards, M.; Scott, M. R.; Goodfriend, E.; Yin, R.; Riston, S.; Ketchesin, K. D.; Moon, H. K.; Petersen, K.; Douaihy, A.; Glausier, J. R.; Brent, D. A.; Lewis, D. A.; Marsland, A. L.; Tseng, G.; Chen, K.; Seney, M. L.; McClung, C. A.; Melhem, N. M.
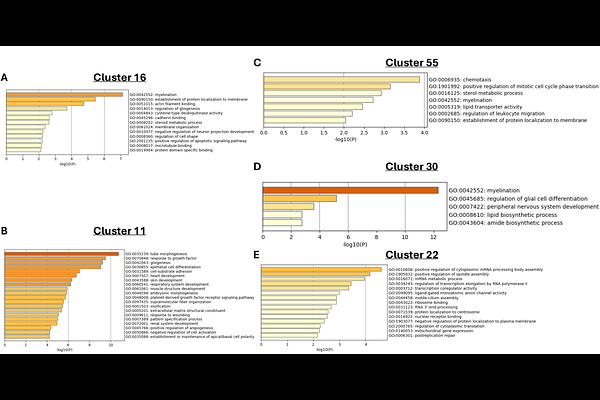
AbstractSuicide is a leading cause of death worldwide, yet the biological mechanisms underlying suicide remain poorly understood. A clearer understanding at the molecular level is essential for developing objective biomarkers and targeted interventions. In this study, we used transcriptomic profiling to investigate gene expression patterns associated with suicidal thoughts and behaviors across peripheral blood (n=264) and postmortem brain tissue from two prefrontal regions (dorsolateral prefrontal cortex, DLPFC; subgenual anterior cingulate cortex, sgACC) of individuals with and without psychiatric illness (n=249). Peripheral analyses revealed broad transcriptional changes associated with suicidal thoughts and behaviors, marked by dysregulated immune-related and inflammatory processes. Longitudinal modeling further revealed gene co-expression modules that predicted future suicide attempts over a 12-month follow-up, highlighting processes related to apoptosis, mitochondrial function, and immune regulation. By contrast, transcriptomic analyses of postmortem tissue derived from the DLPFC and sgACC revealed largely suppressed neuroimmune activity. Gene co-expression analyses in the brain identified suicide-associated modules enriched for synaptic plasticity, oxidative stress, and neuroimmune function, some of which displayed regional specificity. Cross-tissue comparison showed minimal gene-level overlap between brain and blood, although shared pathway-level themes emerged in immune, sensory, and cellular stress processes. Taken together, these findings suggest that suicide is associated with distinct but functionally convergent transcriptional alterations across brain and blood. By integrating tissue-specific and systems-level molecular signatures, this work provides insight into the biological architecture of suicide and lays the groundwork for developing novel biomarkers and therapeutic targets to improve prevention and treatment outcomes.