Mealtime alters daily rhythm in nuclear O-GlcNAcome to regulate hepatic gene expression
Mealtime alters daily rhythm in nuclear O-GlcNAcome to regulate hepatic gene expression
Liu, X.; Cai, Y. D.; Mendiola, A. J. P.; Xu, X.; Luo, Y.; Zheng, H.; Zhao, C.; Chen, C.-H.; Xiang, Y.; Chiu, J. C.
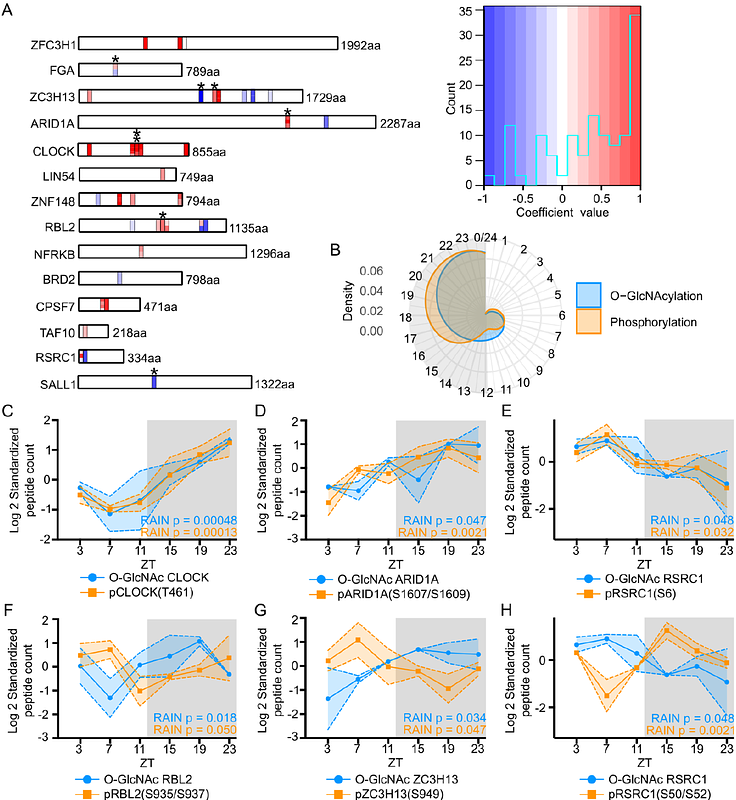
AbstractThe liver circadian clock and hepatic transcriptome are highly responsive to metabolic signals generated from feeding-fasting rhythm. Previous studies have identified a number of nutrient-sensitive signaling pathways that could interpret metabolic input to regulate rhythmic hepatic biology. Here, we investigated the role of O-GlcNAcylation, a nutrient-sensitive post-translational modification (PTM) in mediating metabolic regulation of rhythmic biology in the liver. We observed daily oscillation of global nuclear protein O-GlcNAcylation in the liver of mice subjected to natural nighttime-restricted feeding (NRF). O-GlcNAcomic analysis revealed that 11.54% of 719 O-GlcNAcylated proteins are rhythmically O-GlcNAcylated over a 24-hour day-night cycle. Proteins involved in gene expression were enriched among rhythmically O-GlcNAcylated nuclear proteins, suggesting rhythmic O-GlcNAcylation may directly shape the daily rhythmicity of the hepatic transcriptome. Furthermore, we showed that rhythmic O-GlcNAcylation can also indirectly modulate the hepatic transcriptome by interacting with phosphorylation. Specifically, several proteins harboring O-GlcNAcylation-phosphorylation interplay motif exhibit rhythmic O-GlcNAcylation and phosphorylation. We propose that O-GlcNAcylation may occur at a phospho-degron of a key circadian transcriptional activator, circadian locomotor output cycles kaput (CLOCK), thus regulating its stability and transcriptional output. Finally, we report that unnatural day time-restricted feeding (DRF) dampens O-GlcNAcylation rhythm, suggesting the disruption in liver transcriptomic rhythm previously observed in DRF condition could be mediated by dysregulation of daily protein O-GlcNAcylation rhythm. In summary, our results provide new mechanistic insights into metabolic regulation of daily hepatic transcriptomic rhythm via interplay between O-GlcNAcylation and phosphorylation and shed light on the deleterious effects of improper mealtimes.