Ecological divergence and post-eclosion brain development shape visual performance during Heliconius speciation
Ecological divergence and post-eclosion brain development shape visual performance during Heliconius speciation
Borrero, J.; Alcalde-Anton, A.; Laborieux, L.; Wright, D. S.; Lozano-Urrego, D.; Rueda-Munoz, G.; Pardo-Diaz, C.; Salazar, C.; Montgomery, S. H.; Merrill, R. M.
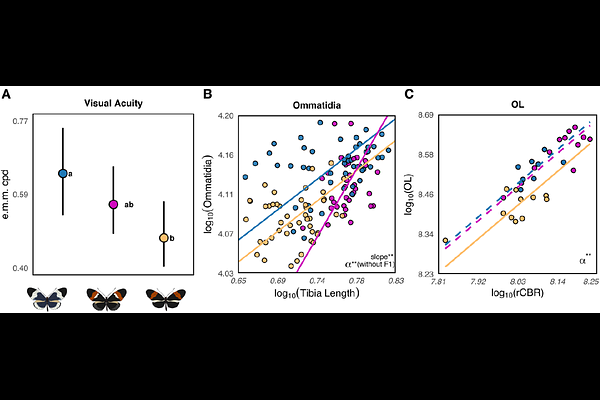
AbstractSensory adaptation is increasingly recognized as a key driver of ecological speciation, but how visual system divergence is coordinated across development, and how this translates into behavioral differences, remains poorly understood. The butterfly Heliconius cydno, which inhabits closed-canopy forests, has larger eyes and greater investment in visual brain centers than its sympatric close-relative H. melpomene, which occupies more open forest-edge habitats, suggesting divergent ecological selection on the visual system. However, the behavioral consequences of these visual adaptations, their developmental trajectories, and whether they break down in hybrids is unknown. To address these questions, we combined ecological field data with behavioral assays and deep-learning-assisted segmentation of neuroanatomy. Visual acuity - the ability to resolve spatial detail - was higher in H. cydno, consistent with its greater ommatidia number, but also increased with age in both species despite no change in external eye morphology. These improvements coincided with the onset of male courtship and female oviposition, suggesting that early adult neurodevelopment shapes visual performance and may support the demands of reproduction. Brain morphology showed species-specific trajectories of post-eclosion optic lobe growth that broadly paralleled increases in acuity and were accompanied by ongoing neurogenesis in the adult optic lobes. While hybrids exhibited intermediate visual acuity, relationships among different components of the visual system were disrupted in hybrids. Together, these results show that ommatidia number alone cannot explain variation in visual acuity, and highlight how coordinated sensory evolution, and its breakdown in hybrids, may contribute to divergence during the early stages of speciation.