Convergent effects of neurodevelopmental disorder-associated variants at mitochondria
Convergent effects of neurodevelopmental disorder-associated variants at mitochondria
Robinette, M. I.; Gundy, J. B.; Leng, X.; Duong, D.; Shantaraman, A.; Shi, L.; Candelario, E. A.; Sing, A.; Garg, S. V.; Niu, W.; Seyfried, N.; Sloan, S. A.; Wen, Z.; Cubells, J. F.; Duncan, E.; Mulle, J. G.; Faundez, V.; Bassell, G. J.; Purcell, R. H.
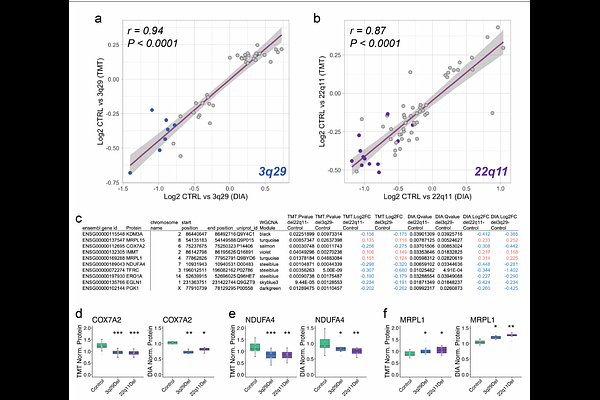
AbstractInvestigations into the molecular pathogenesis of clinically defined neurodevelopmental disorders (NDDs) including autism spectrum disorders (ASD) and schizophrenia (SCZ) have produced evidence implicating dysfunctional mitochondrial metabolism. However, the functional connection between risk variants and mitochondrial proteins largely remains unclear. We tested the hypothesis that proteins encoded by NDD-associated copy number variants (CNVs) and SCZ risk genes are enriched within the mitochondrial interactome. We found that NDD- and SCZ-associated genes exhibit mitochondrial association comparable to their overlap with synaptic proteins, with interaction networks converging most strongly on mitochondrial translation. Two high-risk CNVs, the 3q29 deletion (3q29Del) and the 22q11.2 deletion (22q11Del), confer similar risks for ASD and SCZ and have independently been linked to mitochondrial phenotypes. To test whether these CNVs produce convergent effects on mitochondrial proteins in developing human neural tissue, we generated an isogenic series of 3q29Del and 22q11Del induced-pluripotent stem cells (iPSCs) and differentiated them into forebrain cortical organoids. Quantitative proteomic analysis showed high similarity in the profiles of dysregulated proteins in 3q29Del and 22q11Del compared to isogenic controls. Enrichment analysis of proteins altered in both variants revealed significant convergence on the mitochondrial ribosome and translation machinery. Furthermore, manipulation of mitochondrial translation elicited similar proteomic and functional responses in organoids and neural progenitor cells across both CNVs. These findings indicate that NDD-associated genes have rich interactions with mitochondrial proteins and that two of the strongest risk factors for NDDs may similarly disrupt neural mitochondrial metabolism through impaired mitochondrial translation.