Evolutionary dynamics under phenotypic uncertainty
Evolutionary dynamics under phenotypic uncertainty
Mohanty, V.; Sappington, A.; Shakhnovich, E.; Berger, B.
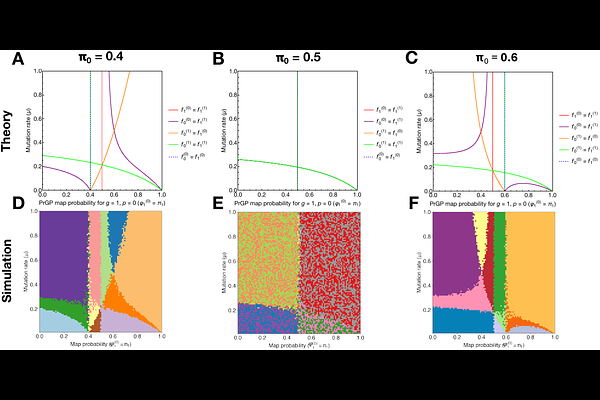
AbstractClassical population genetics has largely relied on the same stochastic differential equations (SDEs) for over 60 years to describe evolutionary dynamics. However, these SDEs ignore the fact that phenotype heterogeneity and noise are ubiquitous in biological systems from bacteria to cancers. Here, we develop Probabilistic Phenotype Genetics (ProP Gen) theory as a mathematical framework for evolutionary dynamics under phenotypic uncertainty. Our newly introduced class of SDEs show that, remarkably, phenotypic uncertainty causes many central tenets of classical population genetics to break down, such as invariance of evolutionary dynamics to global shifts in absolute fitness at fixed population size. We revisit the long-studied problem of valley crossing in rugged fitness landscapes, discovering that low-probability, high-fitness "phenotypic bridges" can substantially accelerate fitness valley crossing even at low mutation rates. We show our theory also explains a paradoxical concept we call "phenotypic buoying" whereby low-fitness phenotypes can exist at surprisingly high frequencies when carried by a high-fitness phenotype which acts as a source. ProP Gen theory uncovers complex phase diagrams of simultaneous coexistence between genotype-phenotype pairs due to phenotypic buoying, which we derive analytically exactly and verify numerically. Notably, the standard Wright-Fisher branching process is unsuitable for probabilistic phenotype numerics because it cannot correctly incorporate phenotypic uncertainty. Thus, we develop a more general, experimentally-inspired discrete-time simulation algorithm, Probabilistic Serial Dilution (ProSeD), which allows for overlapping generations, phenotypic noise, and stochastic phenotype switching. Finally, we show that the new diffusion limit of population genetics recapitulates the experimentally observed resuscitation and partitioning dynamics of bacterial "persister" strains. ProP Gen theory offers promise for describing and predicting the evolutionary dynamics of cancers, which have been empirically observed to exploit phenotypic uncertainty to evade treatment.