HIV-1 Vpr causes separate cell cycle arrests in G2 and M that activate alternative DNA damage pathways
HIV-1 Vpr causes separate cell cycle arrests in G2 and M that activate alternative DNA damage pathways
Hall, R.; Ahern, L. M.; Yap, M. W.; Tsai, M.-H. C.; Boucherit, V. C.; Takaki, T.; Boulton, S. J.; Bishop, K. N.
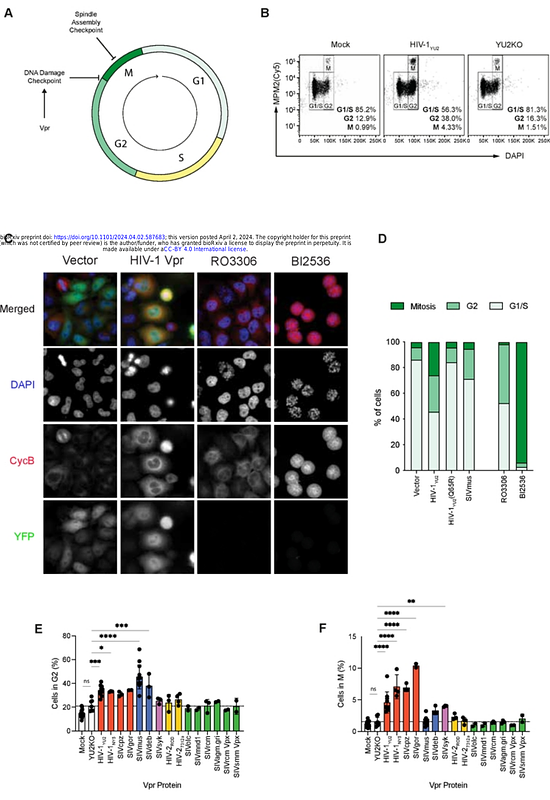
AbstractVpr is a conserved primate lentiviral accessory protein that induces cell cycle arrest in G2. The precise mechanism of this arrest and its benefit to viral replication is unknown. Here, we show that in addition to G2 arrest, Vpr from HIV-1/SIVcpz and HIV-2 lineages separately induce mitotic arrest through the spindle assembly checkpoint, in contrast to other Vpr proteins that only cause G2 arrest. The G2 arrest was mediated solely by ATR (ataxia telangiectasia and Rad3 related) and this activity caused elevated cellular dNTP levels. The mitotic arrest required ATM (ataxia-telangiectasia mutated) as well as ATR activity and resulted from the formation of HIV-1 Vpr-induced ultra-fine anaphase bridges. Moreover, ectopic expression of the DNA structure-specific endonuclease, MUS81, prevented mitotic but not G2 arrest. Importantly, virion-incorporated Vpr was sufficient to induce cellular changes within 12h post-infection, implying that these events benefit the early stages of HIV infection.