DNA Damage Driven Viability Loss and Transcriptional Reprogramming in Chinese Hamster Ovary Cell Perfusion Culture
DNA Damage Driven Viability Loss and Transcriptional Reprogramming in Chinese Hamster Ovary Cell Perfusion Culture
Hitchcock, N. B.; Annoh, M.; Grassi, L.; Das, S.; Sayago Ferreira, C.; Ray, D.; Elgendy, R.; Wang, L.; Lee, K.; Sudbery, I. M.; Bose, D. A.; Hatton, D.; Sou, S. N.; Mistry, R.; Toseland, C. P.
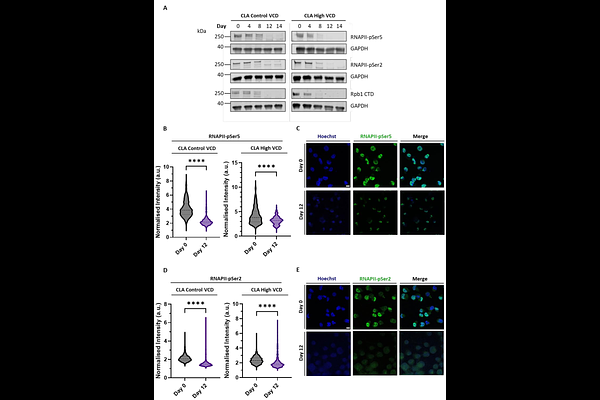
AbstractIntensified perfusion cultures promise higher yields and consistent product quality, yet extended runs frequently stall due to declining Chinese hamster ovary (CHO) cell viability at high cell density. Here, we identify DNA damage accumulation as a central, previously underappreciated driver of this limitation. Using two antibody-expressing CHO cell lines operated at a control and high cell density in perfusion bioreactors, we combined time-resolved transcriptomics with molecular, biophysical and super-resolution imaging analyses. We observed a progressive, global downregulation of DNA damage response (DDR) pathways accompanied by a time-dependent accumulation of DNA lesions. Notably, {gamma}H2AX signalling declined despite DNA damage, indicating impaired damage sensing and repair. Concomitantly, RNA polymerase II protein levels and transcriptional hub organisation were markedly reduced, consistent with widespread transcriptional dysfunction preceding loss of viability. An extended 21-day perfusion run confirmed continued viability decline beyond day 14, supporting a cumulative damage model. Comparison with HEK293 cells revealed substantially more efficient DNA repair, underscoring an intrinsically attenuated DDR in CHO cells. Together, these findings demonstrate that unrepaired DNA damage fundamentally constrains perfusion culture longevity, exposing limits of CHO genomic plasticity and highlighting DDR pathways as promising targets for host cell engineering to enhance perfusion performance.