A spatial-temporal atlas of human islet pathophysiology identifies a size-dependent trajectory from compensation to decompensation
A spatial-temporal atlas of human islet pathophysiology identifies a size-dependent trajectory from compensation to decompensation
LIU, T.; Liang, R.; ZHANG, L.; Ma, W.; WANG, X.; Ren, H.; Wang, S.
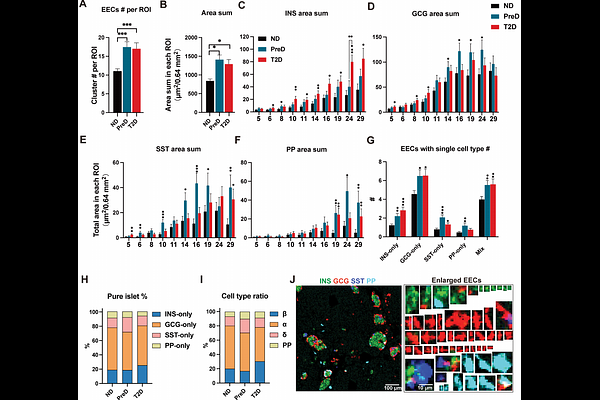
AbstractType 2 diabetes (T2D) is characterized by progressive islet dysfunction, yet the transition from functional adaptation to failure remains poorly defined within the native tissue architecture. Using multiplex imaging mass cytometry, we systematically analyzed human pancreatic islets across a spectrum of non-diabetic (ND), prediabetic (PreD), and T2D donors, revealing that pathological remodeling is profoundly size-dependent. This remodeling is manifested as a coordinated evolution of subpopulation abundance, endocrine cell proportions, structural integrity, and protein expression profiles, revealing that islets of different sizes undergo divergent fates during disease progression. We identified a size-dependent vulnerability spectrum where medium-and large- sized islets (>100 um in diameter) serve as the primary histopathological correlates of glycemic failure (HbA1c), exhibiting early density loss and structural disintegration. In contrast, small islets (30-100 um in diameter) exhibited compensatory hormone upregulation during PreD. Notably, diverging from the 'death of the small' paradigm in Type 1 Diabetes, our data reveal a significant expansion of extra-islet endocrine clusters (EECs) that initiates during the compensatory stage, effectively preceding the onset of overt hyperglycemia. Finally, t-SNE clustering reconstructed a continuous trajectory of islet remodeling, capturing the phenotypic evolution of islets from normoglycemia through compensation to clinical decompensation. This study provides a high-resolution atlas of islet pathophysiology, offering new insights into T2D progression.