Physical continuity at biomaterial-ECM interfaces regulates fibroblast activation via NF-κB
Physical continuity at biomaterial-ECM interfaces regulates fibroblast activation via NF-κB
Suarez-Arnedo, A.; Harris, M.; Robinson, C.; Riley, L.; Kim, A.; Zhang, L.; Hoffman, B. D.; Segura, T.
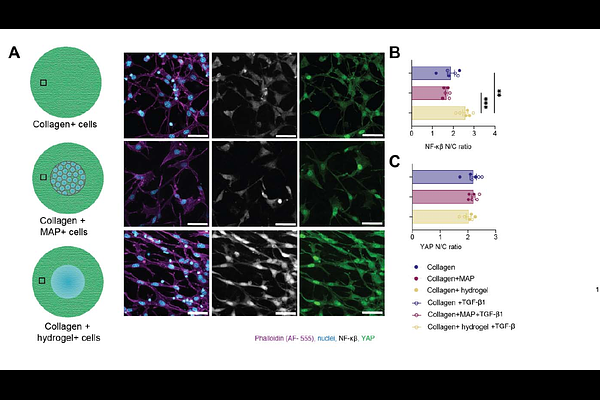
AbstractFibrotic responses at biomaterial-tissue interfaces limit implant integration and regenerative healing, yet how the interaction between biomaterials and the extracellular matrix (ECM) regulates fibroblast activation remains poorly understood. Granular hydrogels including microporous annealed particle scaffolds (MAP) reduce fibrosis, while chemically and mechanically matched hydrogels do not, suggesting a dominant role for scaffold architecture. In this model, MAP scaffolds allow collagen infiltration and form physically continuous composites, whereas hydrogels exclude collagen and generate interfacial slip planes. To isolate how biomaterial architecture influences extracellular matrix (ECM) integration and fibroblast activation, we developed a reductionist in vitro model that integrates collagen type I with either microporous annealed particle (MAP) scaffolds or chemically and mechanically matched bulk hydrogels. This physical integration stabilizes collagen architecture, limits fibroblast-mediated matrix compaction, suppresses contractility, and attenuates myofibroblast transition. Fibroblasts in mechanically integrated environments exhibit reduced expression and nuclear localization of NF-KappaB and are enriched for quiescent phenotypes. Together, these findings identify biomaterial-ECM physical continuity as a design principle for limiting fibrotic signaling.