The broad-spectrum RumC1 bacteriocin targets a transient peptidoglycan intermediate of the nascent cell wall
The broad-spectrum RumC1 bacteriocin targets a transient peptidoglycan intermediate of the nascent cell wall
BOYELDIEU, A.; BERGE, M.; ROBLIN, C.; SHAMSEDDINE, L.; DIAZ-ROVIRA, A. M.; SOULET, A.-L.; BASSET, C.; PLOUHINEC, L.; AMOURIC, A.; MILHAVET, S.; PERRAULT, L. M.; MARX PEREIRA DA CUNHA, J.; JOHNSTON, C.; KIEFFER-JAQUINOD, S.; MARESCA, M.; PERRIER, J.; CHOUQUET, A.; MORLOT, C.; GUALLAR, V.; LAFOND, M.; DUARTE, V.; Campo, N.; Polard, P.
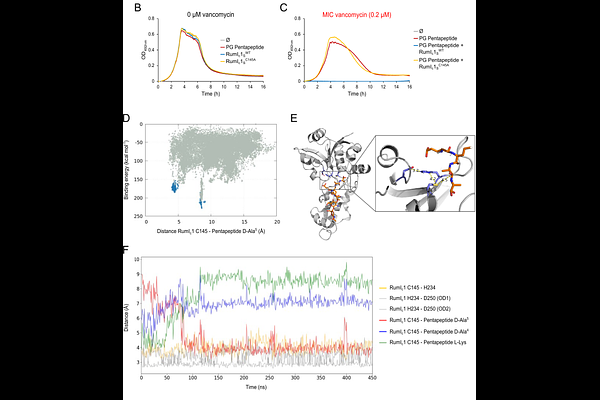
AbstractRumC1 is a structurally unique bacteriocin with broad-spectrum efficacy, including against multidrug-resistant pathogens, yet acting by an undefined mechanism. By integrating genetics, biochemistry, computational modeling and single-cell fluorescence microscopy, we demonstrate that RumC1 is a distinct cell-wall-targeting toxin. First, all RumC1-resistant mutants isolated through a high-rate, genome-wide mutagenic screening exhibited specific impairments in peptidoglycan homeostasis regulation, pinpointing this pathway as critical for RumC1 activity. Second, RumC1 selectively accumulates within neosynthesized peptidoglycan, leading to cell growth arrest and death in a dose-dependent manner. Third, we characterize the RumIc1 immunity protein of the RumC1 biosynthetic cluster as a peptidase acting at the cell surface to protect the cells by trimming the stem peptide crucial for cell-wall assembly. As such, RumIc1 provides cross-protection against vancomycin, while RumC1 is demonstrated to act differently from this glycopeptide antibiotic. Collectively, these findings establish RumC1 as a toxin targeting a key peptidoglycan intermediate of cell wall maturation.