The pangenome of Aspergillus fumigatus highlights the dynamics of gene gain-loss over evolutionary timescales in a human fungal pathogen
The pangenome of Aspergillus fumigatus highlights the dynamics of gene gain-loss over evolutionary timescales in a human fungal pathogen
Chown, H.; Rhodes, J.; Fisher, M. C.; Bromley, M. J.
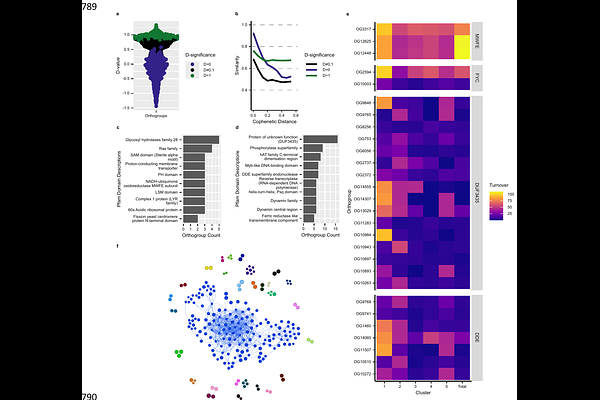
AbstractHow fungal pathogens generate and maintain genetic diversity under sustained environmental drug pressure is central to understanding the emergence of antifungal resistance. In environmental moulds such as Aspergillus fumigatus, escalating exposure to agricultural fungicides that share targets with clinical azoles imposes chronic selection outside the host, yet the genomic mechanisms enabling long-term adaptation remains unclear. Here, we reconstruct the largest eukaryotic pangenome assembled to date, comprising over 1,000 A. fumigatus isolates collected across 34 countries spanning a century. Using network-based orthogroup clustering combined with ancestral state reconstruction, we show that the A. fumigatus pangenome is open and shaped by continual gene gain and loss. Pangenome-wide association analyses identify accessory genes associated with itraconazole resistance, indicating that resistance evolution occurs within broader genomic backgrounds and beyond canonical target-site mutations. We further reveal that the accessory genome is structured into distinct evolutionary cohorts, including lineage-restricted gene sets enriched for mobile genetic element-associated domains, notably Starship-linked genes. These patterns suggest that Starships contribute to clade-specific genome architecture while remaining largely constrained by phylogenetic boundaries. Time-calibrated phylogenetic modelling reveals a relatively slow rate of gene turnover - approximately two orthogroup events per century - demonstrating that large-scale genome evolution in A. fumigatus is decoupled from elevated point mutation rates and contrasts sharply with bacterial systems. Together, these findings establish a quantitative framework for fungal pangenome evolution and reveal how structured accessory genome dynamics underpin antifungal resistance and long-term adaptation in this major human pathogen.