Dopamine vesicles are specified by mechanisms overriding canonical synaptic vesicle size constraints
Dopamine vesicles are specified by mechanisms overriding canonical synaptic vesicle size constraints
Fujise, K.; Karmali, N.; Mishra, J.; Kimura, H.; Tsuji, T.; Saito, A.; Mohd Rafiq, N.
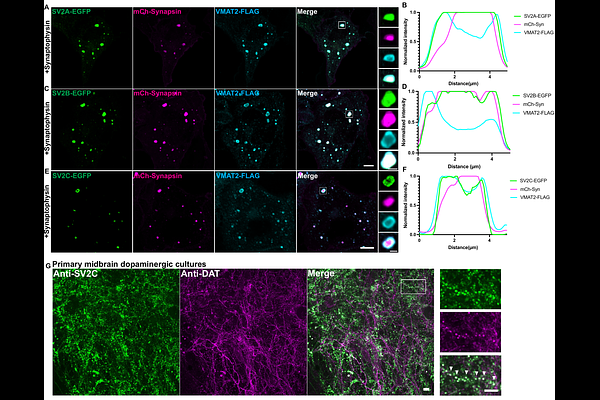
AbstractDopaminergic neurons contain distinct synaptic vesicle (SV) populations that segregate dopamine from glutamate, but the molecular basis of this segregation remains unclear. Using an established in vitro SV reconstitution system in fibroblasts, we show that synaptogyrin family proteins - synaptogyrin 1, synaptogyrin 2 and synaptogyrin 3 - but not synaptophysin, selectively associate with VMAT2-positive dopamine vesicles in the presence of synapsin. Despite the known role of synaptophysin and synaptogyrin family proteins in generating small SVs (40-50 nm), VMAT2 vesicles retain a larger size (50-80 nm) when co-expressed with synaptogyrins, while remaining segregated from synaptophysin-containing vesicles. SV2 family proteins (SV2A, SV2B, and SV2C) localize to both VMAT2- and synaptophysin-positive vesicle populations, with SV2C preferentially enriched on VMAT2-positive dopamine vesicles. Consistent with the selective dystrophy of SV2C/VMAT2/DAT-positive dopaminergic terminals in parkinsonism-associated SJ1RQKI mice, iPSC-derived dopaminergic neurons carrying the same mutation accumulate small and enlarged SV-like organelles within autophagosomes, indicating defective SV clearance. Together, our findings suggest that dopamine vesicles are regulated by distinct trafficking mechanisms that override canonical SV size control, providing insight into the selective vulnerability of dopaminergic terminals in Parkinson's disease.