Comprehensive analysis of air and surface hospital microbiomes uncovers potential hotspots and avenues for transmission of diverse pathogens linked to hospital-acquired infections
Comprehensive analysis of air and surface hospital microbiomes uncovers potential hotspots and avenues for transmission of diverse pathogens linked to hospital-acquired infections
Cambara, J. C. O.; Previtali, O.; Cuber, P.; Lebre, P. H.; Ali, S.; Chooneea, D.; Misra, R.; Mkrtchyan, H. V.
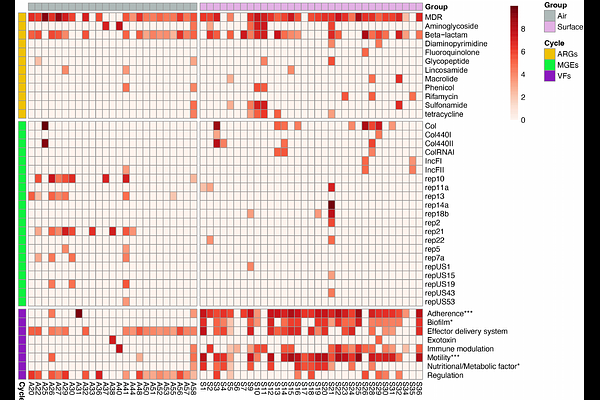
AbstractFomite-mediated and airborne transmission pathways play a significant role in the dissemination of healthcare associated pathogens, contributing to the burden of hospital-acquired infections (HAIs). Effective infection prevention and control therefore require robust surveillance approaches capable of capturing the complexity of air and surface microbial communities present in healthcare settings. However, to date such approaches are not widely employed. In this study we used a rapid and low-biomass optimized metagenomic workflow to detect clinically relevant pathogens, characterise genetic signatures such as antimicrobial resistance and virulence determinants, and profile the wider airborne microbiome, including unculturable taxa) essential for HAI surveillance. Air samples were collected from multiple clinical and non-clinical areas across three multi-storey healthcare units within a UK hospital, alongside to surface samples from a haematology/oncology ward with a documented history of outbreaks. Following DNA extraction and enrichment optimised for low-biomass samples, sequencing was performed using the Oxford Nanopore Technologies MinIon long-read platform. Metagenomics data were analysed using an in-house developed bioinformatics pipeline. Metagenomic profiling revealed high bacterial taxonomic diversity across sampled environments, with limited overlap between the airborne and surface communities. Approximately 0.3% of bacterial reads harboured a large variety of antimicrobial resistance genes (ARGs), virulence factors (VFs) and mobile genetics elements (MGEs) across different hospital sample groups. Notably, critical and emerging pathogens were detected across ten wards and were associated with clinically significant resistance determinants, including multiple blaOXA subtypes, vanA gene clusters, and multidrug efflux pumps conferring resistance to eight important classes of antibiotics, including carbapenems, cephalosporins, penams, and vancomycin. In addition, several plasmid replicons implemented in horizontal gene transfer (HGT) were identified within these pathogens, indicating an increased potential for the emergence and dissemination of multi-drug resistance within hospital associated microbial communities. Our findings highlight the importance of using rapid metagenomics-based methodologies for environmental surveillance in healthcare settings. Correspondingly, this study revealed that hospital air and surface microbiomes comprise complex and dynamic microbial communities that harbour diverse of ARGs and VFs with the potential for rapid transmission across the surface-air interface, particularly in high-risk/vulnerable patient areas where outbreaks are more likely to occur.