Single-cell carbon storage dynamics drive conditional fitness in microbes
Single-cell carbon storage dynamics drive conditional fitness in microbes
Huang, J.; Yuan, R.; Ma, Y.; Ma, H.; Arkin, A. P.
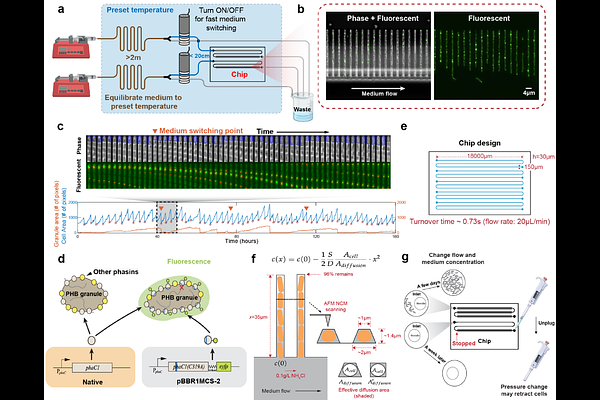
AbstractMicrobes frequently encounter fluctuating environments, requiring dynamic energy management strategies for survival. While carbon storage polymers like polyhydroxybutyrate (PHB) are ubiquitous across bacterial taxa, their precise ecological advantage remains poorly understood (1). Here we show that carbon storage drives conditional fitness during environmental transitions. Using a high-throughput single-cell microfluidic platform, we tracked tens of thousands of Cupriavidus necator cells under precisely controlled carbon and nitrogen fluctuations. We found that PHB provides no advantage under nutrient abundance but becomes decisive at starvation boundaries: during carbon starvation, it enables ~30% more progeny before arrest; during recovery from nitrogen starvation, it shortens lag and accelerates regrowth. Strikingly, at the single-cell level, PHB granules are inherited in an asymmetric, all-or-nothing fashion, concentrating resources into specific lineages to overcome the discrete energetic threshold required for cell division. Despite this single-cell variance, at the population level, PHB fractions robustly return to a common setpoint after nutrient shifts - a homeostatic behavior consistent with integral feedback control. These findings reveal that while PHB does not increase the basal exponential growth rate, it confers a distinct fitness advantage by prolonging the proliferative phase during nutrient depletion and facilitating successful recovery from starvation, explaining the evolutionary persistence of carbon storage in environments with pulsed resource availability.