A Seven-Protein Assembly Promotes Stability, Neutralisation and Secretion of the T7SSb LXG-effector TelE
A Seven-Protein Assembly Promotes Stability, Neutralisation and Secretion of the T7SSb LXG-effector TelE
Agrawal, P.; Gonet, A.; Toko, D.; Gorkisch, J.; Raoux-barbot, D.; du Merle, L.; Bouscasse, E.; Matondo, M.; Mechaly, A.; Keong Teh, W.; Bezault, A.; Chenal, A.; Bellinzoni, M.; Dramsi, S.; Gubellini, F.
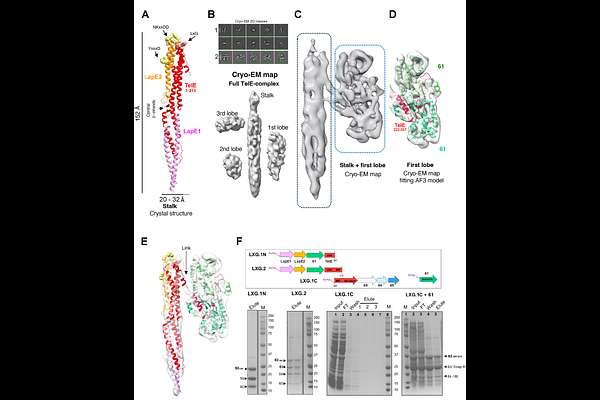
AbstractStreptococcus gallolyticus subsp. gallolyticus (SGG) is a gut pathobiont associated with colorectal cancer. Like many Firmicutes, SGG utilizes a specialized Type VII Secretion System (T7SSb) to export WXG100 and LXG proteins, the latter involved in bacterial competition. We previously identified TelE, an LXG protein whose C-terminus mediates membrane pore formation in Escherichia coli. In SGG UCN34, TelE (Gallo_0562) is co-expressed with six proteins (Gallo_0559 to Gallo_0565), including its immunity protein TipE (Gallo_0565). Here we show that the absence of those co-expressed proteins affects TelE stability and secretion. These proteins associate with TelE to form a soluble and stable seven-protein complex. Gallo_0559 and Gallo_0560 interact with the N-terminal LXG domain of TelE, Gallo_0561 binds to its central region, while the six-transmembrane protein Gallo_0563, together with Gallo_0564 and TipE associates with its C-terminal domain. These findings describe a new modular complex that stabilizes TelE while reducing its toxicity and optimizing its T7SSb-mediated delivery.