Recessive POPDC1 Truncation Causes Lethal Short-QT Pattern Arrhythmogenic Cardiomyopathy with Multi-Ion Channel Remodeling and Ankyrin-G Scaffold Disruption
Recessive POPDC1 Truncation Causes Lethal Short-QT Pattern Arrhythmogenic Cardiomyopathy with Multi-Ion Channel Remodeling and Ankyrin-G Scaffold Disruption
luo, R.; Zheng, C.; Lan, H.; He, Y.; Wang, Y.; Sheng, Q.; Li, S.; Deng, H.; Yao, L.; Li, Y.; Lim, W.-W.; Hua, W.; Wu, X.; Li, X.
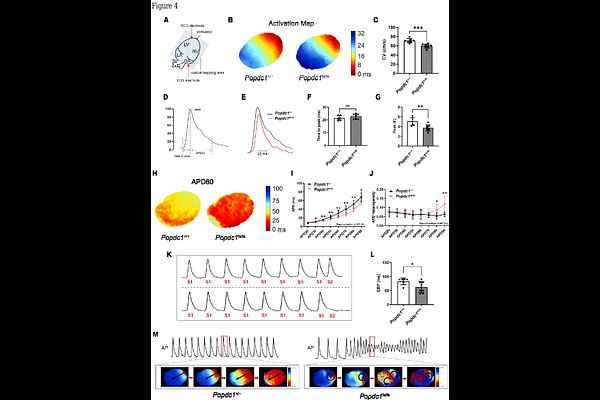
AbstractAbstract Aims: Biallelic variants in Popeye domain containing 1 (POPDC1) classically cause limb-girdle muscular dystrophy, but their impact on cardiac system remains unclear. We investigated the functional consequence of a POPDC1 frameshift variant (c.448delT), first identified in a consanguineous family with arrhythmogenic cardiomyopathy (ACM) and sudden death. Methods and results: Comprehensive clinical and genetic evaluation was followed by mechanistic studies in an orthologous Popdc1 knock-in rat model. Functional characterization included biotelemetry and programmed electrical stimulation, optical mapping, patch clamp, intracellular Ca2+ imaging, proteomics, and oxidative stress assays. Homozygous mutants (Popdc1fs/fs) exhibited a shortened QTc interval and high incidence of ventricular tachycardia compared to wild-type (Popdc1+/+). Urethane anesthesia provoked second-degree AV block in all 10 Popdc1fs/fs rats but in only 1 of 9 Popdc1+/+ littermates. Optical mapping demonstrated abbreviated action potentials, slowed conduction velocity, and inducible polymorphic VT. Patch clamp confirmed accelerated repolarization, with upregulated transient outward potassium currents (Ito) and L-type calcium currents (ICa, L), but downregulated peak sodium currents (INa). Multi-omics and ultrastructural analyses revealed a post-translational collapse of intercalated disc: POPDC1 loss destabilized Ankyrin-G and its membrane-anchored cargo, Nav1.5, coinciding with enhanced Kv4.3 and Cav1.2 protein. These disruptions create a convergence of delayed conduction and shortened refractoriness, forming substrate for malignant re-entry. Conclusion: We defined a recessive short-QT ACM caused by biallelic POPDC1 truncation driving Ankyrin-G disruption, manifesting as a triad: (1) bradycardia/AV block, (2) accelerated repolarization as short-QT pattern/diffuse T-wave flattening or inversion, and (3) progressive cardiomyopathy and sudden death.