Streptomyces enrichment in roots during drought is uncoupled from plant benefit and is driven by host suppression of iron uptake and immunity
Streptomyces enrichment in roots during drought is uncoupled from plant benefit and is driven by host suppression of iron uptake and immunity
Fitzpatrick, C.; Smith, R.; Hige, J.; Law, T.; Russ, D.; Ajayi, O.; Eida, A.; Jacob, P.; Jowers, M.; Kumar, N.; Lai, C.; Anguita-Maeso, M.; Peterson, B.; Saha, C.; Skelly, T.; Zhao, Q.; Zhou, W.; Grant, S.; Mougous, J.; Jones, C.; Dangl, J.
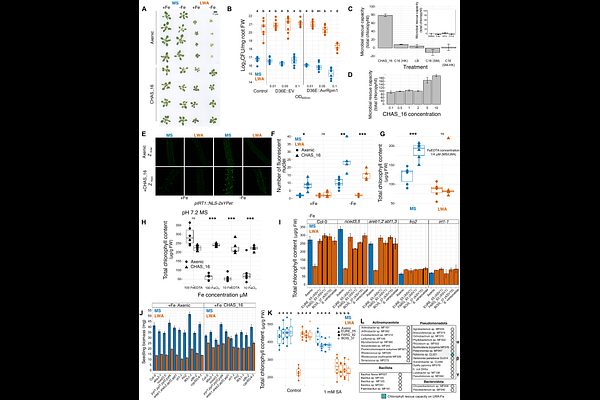
AbstractDrought reshapes plant root microbiota, yet the mechanistic drivers and consequences of this observation remain unclear. We discovered that suppression of host immunity and iron homeostasis is required for Streptomyces enrichment in roots during drought across diverse soils. Genetic and physiological manipulation of these host pathways confirmed their requirement in modulating Streptomyces root enrichment. Drought-induced suppression of iron uptake was conserved across the ~160 My monocot-eudicot divergence. Some Streptomyces strains enhanced plant growth and rescued iron uptake under drought. These benefits were uncoupled from Streptomyces root enrichment. They were instead shaped by intra-Streptomyces antagonism. We propose a two-step model: drought-driven down regulation of host iron and immune pathways enriches Streptomyces, while intra-genus dynamics fine-tune strain-level assembly and functional outcomes. Our data refine the idea that Streptomyces are enriched in roots during drought in response to a plant cry for help and consequently contribute to alleviation of this abiotic stress.