Lmna deficiency promotes EPHX2 nuclear translocation to ameliorate cardiac dysfunction in mice
Lmna deficiency promotes EPHX2 nuclear translocation to ameliorate cardiac dysfunction in mice
Guo, C.; Wang, Z.; Liu, J.; Wu, C.; Yang, Y.; Lv, Z.; Chen, G.; Sun, Y.; Bai, R.; Sun, W.; Lu, T.; Wang, K.; Tian, Z.; Zhang, X.; Zhao, D.; Zhang, S.; Guo, Y.
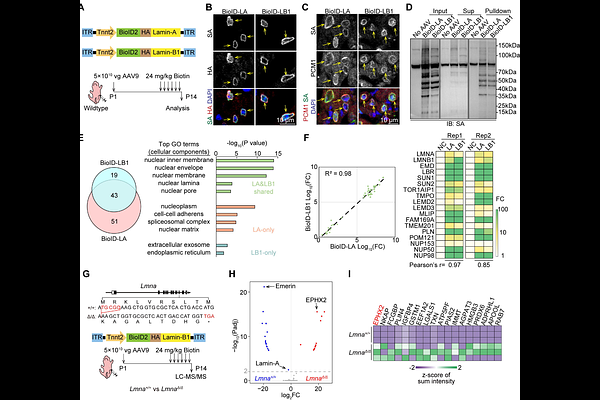
AbstractBackground Cardiovascular diseases are often associated with altered protein subcellular localization. As a major cause of inherited cardiomyopathy, LMNA deficiency could trigger nuclear envelope rupture and broadly impair the localization of nuclear and cytoplasmic proteins. Systemic approaches to identify, dissect and manipulate the localization of endogenous proteins are important for mechanistic and therapeutic investigation. Method Proximity proteomics of the nuclear lamina was performed specifically in cardiomyocytes in Lmna-deficient murine models. AAV-mediated Cas9-based gene silencing and subcellular gene upregulation were conducted via the nuclear localization signal (NLS) and nuclear export signal (NES). Cas9-based somatic mutagenesis was supplemented with the single-strand DNA templates of AAV to achieve robust homology-directed repair (HDR) and targeted NLS knock-in, which translocated cytoplasmic proteins into nuclei. Result In vivo proximity proteomics detected increased epoxide hydrolase 2 (EPHX2) in cardiomyocyte nuclei in mice carrying germline or cardiac-specific Lmna truncating variants. This phenotype was associated with ruptured nuclear envelope. Cas9-mediated Ephx2 knockout in cardiomyocytes ameliorated cardiac dysfunction in Lmna-deficient mice. Strikingly, overexpression of NLS-EPHX2, but not NES-EPHX2, also mitigated cardiac dysfunction. The cardiac protective EPHX2 substrates, epoxyeicosatrienoic acids (EETs), did not alter upon NLS-EPHX2 overexpression. By contrast, the Lmna-related DNA damage marker {gamma}-H2AX was reduced. The EPHX-D333A mutant lacking hydrolase activity recapitulated the effects of wildtype EPHX2 in nuclei. AAV-Cas9-based HDR achieved efficient NLS knock-in and EPHX2 nuclear translocation in more than 60% cardiomyocytes, which improved cardiac function. Conclusion Lmna deficiency leads to the nuclear translocation of EPHX2, which ameliorated cardiac dysfunction in a hydrolase-independent manner. AAV-HDR-mediated somatic gene editing provides an efficient approach to manipulate the subcellular localization of endogenous proteins in cardiomyocytes in vivo.