Brca1 heterozygosity leads to hepatic steatosis in male and female mice despite sexually dimorphic effects on systemic metabolism
Brca1 heterozygosity leads to hepatic steatosis in male and female mice despite sexually dimorphic effects on systemic metabolism
Palikhe, S.; Qiao, L.; Kutz, C.; Liang, X.; Dhorajiya, P.; Koestler, D. C.; Bhardwaj, P.; McCoin, C. S.; Peterson, O.; Tse, H. M.; Thyfault, J. P.; Brown, K. A.
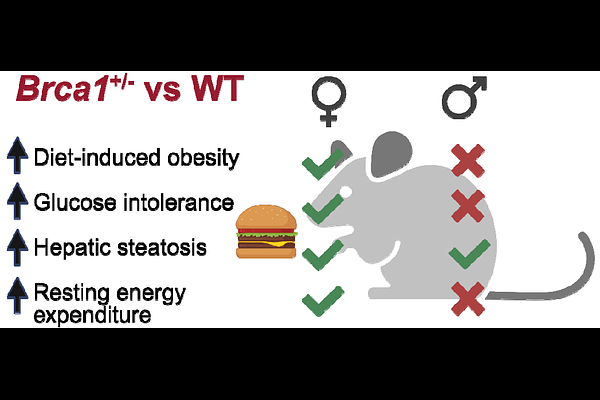
AbstractCarrying a germline mutation in BRCA1 is associated with an increased risk of several cancers, including breast and ovarian. Our recent work has demonstrated that obesity is associated with elevated levels of DNA damage in breast glands in this high-risk population. BRCA1 is a canonical tumor suppressor gene primarily recognized for its role in DNA damage repair, yet emerging evidence suggests broader functions in metabolic regulation. To determine whether heterozygous loss of Brca1, as seen in individuals who carry a germline mutation, modifies susceptibility to diet-induced metabolic dysfunction in a sex-dependent manner, we subjected wild-type (WT) and Brca1+/- mice of both sexes to a high-fat diet (HFD) and performed longitudinal metabolic phenotyping. Female Brca1+/- mice exhibited pronounced obesity, increased adiposity, hyperinsulinemia, and impaired glucose tolerance. In contrast, male Brca1+/- mice showed modest resistance to HFD-induced weight gain and displayed improved glucose tolerance compared to WT controls. Notably, Brca1 heterozygosity led to more severe hepatic steatosis with HFD, indicating a shared susceptibility to liver lipid accumulation despite divergent systemic outcomes. In females, steatosis was associated with reduced mitochondrial respiratory complex IV activity and transcriptional remodeling that favored lipid storage. Treatment with the dual GLP-1/GIP receptor agonist tirzepatide ameliorated systemic metabolic dysfunction and hepatic steatosis in HFD-fed female Brca1+/-; mice. These findings identify Brca1 heterozygosity as a modifier of metabolic disease risk, expanding BRCA1 biology beyond tumor suppression.