Prophase Chromosomes Relocalizes to Nuclear Periphery for Protection in Depletion of Nucleoporin NPP-3/NUP205 Through the Spindle Assembly Checkpoint Activity, Centromere-Kinetochore Proteins and BAF-1-LEM-2
Prophase Chromosomes Relocalizes to Nuclear Periphery for Protection in Depletion of Nucleoporin NPP-3/NUP205 Through the Spindle Assembly Checkpoint Activity, Centromere-Kinetochore Proteins and BAF-1-LEM-2
Jiang, L.; Tse, Y. C.; Yuen, K. W. Y.
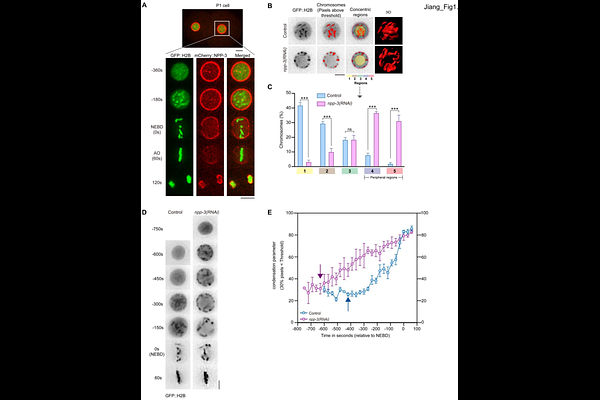
AbstractThe nuclear envelope (NE) mediates transport between nucleus and cytoplasm in eukaryotic cells and protects genetic materials against cytoplasmic enzymes. Nuclear pore complexes (NPCs) regulate chromosome architecture, genome integrity, gene transcription, and cell division. In Caenorhabditis elegans embryos, depletion of NPP-3/NUP205 causes NE rupture, premature chromosome condensation, and relocalization of condensed chromosomes to the nuclear periphery, similar to responses during anoxia and quiescence. This chromosomal relocalization depends on the spindle assembly checkpoint (SAC), inner kinetochore proteins, and partially on the NE rupture repair proteins BAF-1 and LEM-2. NPP-3 depletion prolongs prophase and prometaphase, as mediated by SAC proteins MDF-1 and MDF-2. Additionally, NPP-3 depletion alters MDF-1 localization, removing it from NE and increasing its nuclear accumulation, while reducing import of kinetochore components such as KNL-1, BUB-1, and HCP-1. In 20-30 cell-stage embryos, MDF-1 foci are observed on peripheral chromosomes during prophase. Both MDF-1 and MDF-2 accumulate on chromosomes during prometaphase. The increased incidence of lagging chromosomes, DNA damage, and micronuclei upon NPP-3 and MDF-1 depletion, suggesting that peripheral chromosome localization may serve as a protective mechanism against DNA damage. These findings shed light into cellular responses to NE rupture, with potential implications for laminopathies and cancers involving nuclear envelope defects.