Inhibition of Acid Sphingomyelinase Links Sphingolipid Remodeling to Necroptotic Cell Death
Inhibition of Acid Sphingomyelinase Links Sphingolipid Remodeling to Necroptotic Cell Death
Pilapil, L.; Chitkara, S.; Atilla-Gokcumen, G. E.
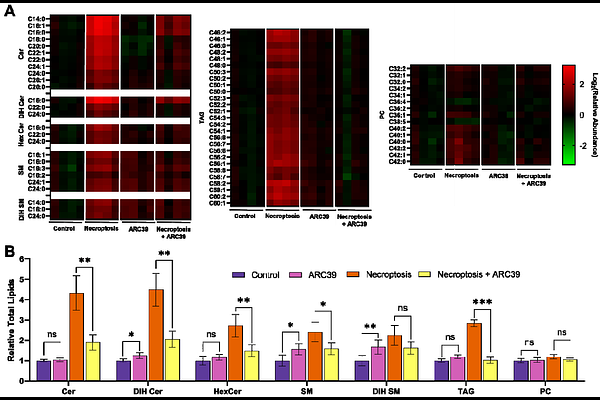
AbstractNecroptosis is a lytic form of programmed cell death that requires activation of the RIPK1/3 and MLKL complex and results in plasma membrane permeabilization. Although the protein components governing necroptosis are well defined, the lipid determinants of this process remain poorly understood. Here, we combined lipidomics, pharmacological perturbations of sphingolipid metabolism and functional assays to identify sphingolipid pathways that contribute to necroptotic cell death. Using a panel of small molecule inhibitors, we found that inhibition of acid sphingomyelinase (ASMase) with ARC39 restored cell viability and membrane integrity during necroptosis without altering canonical necroptotic signaling. Lipidomic analysis revealed that ARC39 treatment prevented ceramide accumulation in necroptosis, linking reduced ceramide levels to decreased membrane permeability. Interestingly, ARC39 treatment did not reduce total cellular levels of phosphorylated MLKL (pMLKL) nor its initial membrane association, suggesting that the observed decrease in membrane permeability arises downstream of MLKL activation. Instead, our findings support a model in which the reduction of ceramide levels impairs productive membrane insertion and pore formation by pMLKL. Consistent with this interpretation, genetic knockdown of ASMase similarly resulted in increased cell viability, decreased membrane permeabilization, and decreased ceramide levels during necroptosis, further linking ceramide homeostasis to necroptotic membrane damage. Together, these results indicate that ASMase-derived ceramides are important for efficient MLKL-mediated membrane permeabilization in necroptosis.