Integrated Skin Sensitization Assessment Using Consensus Modelling and Tiered New Approach Methodology: A SaferSkin Case Study
Integrated Skin Sensitization Assessment Using Consensus Modelling and Tiered New Approach Methodology: A SaferSkin Case Study
Hardy, B.; Mohoric, T.; Parween, S.; Lopez, B.; Daligaux, P.; Darde, T.; Chesne, C.; Stockman, N.; Lemos, J.; Saiakhov, R.; Boglari, C.; Poon, A.; Ukaegbu, D.; Andrade, C.
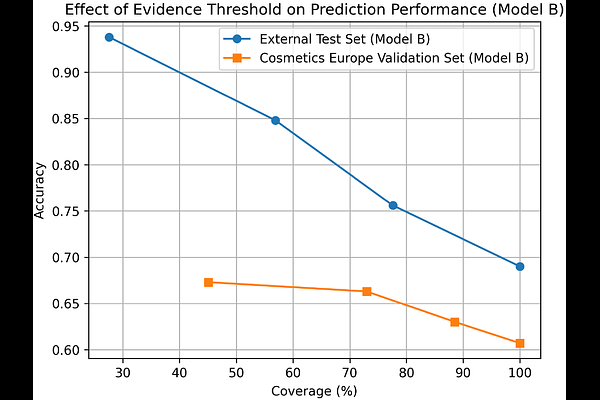
AbstractThe transition toward animal-free safety assessment of chemicals has accelerated the development of New Approach Methodologies (NAMs) for predicting skin sensitization. However, individual in silico models and experimental NAM assays frequently produce inconsistent or contradictory results, limiting their reliability when used in isolation. To address this challenge, we present a tiered integrated assessment framework implemented through the open source SaferSkin application, which enables systematic comparison and integration of multiple predictive models and experimental data within a transparent weight-of-evidence workflow. In this case study, a diverse set of 21 reference compounds was evaluated using a battery of in silico approaches, including the OECD QSAR Toolbox, VEGA, CASE Ultra and additional machine-learning models implemented within SaferSkin. The platform enables side-by-side comparison of predictions and integration of experimental data through Bayesian network models, allowing probabilistic updating of predictions as new evidence becomes available. Our results demonstrate that reliance on any single predictive model is insufficient for robust hazard identification due to frequent disagreement between models. In contrast, consensus interpretation across multiple modelling approaches combined with targeted experimental evidence substantially improves predictive confidence. The integrated weight-of-evidence framework showed strong concordance with reference classifications and was further supported by independent validation using the Pred-Skin Bayesian model. Importantly, the tiered workflow enables resolution of ambiguous cases. For example, lower-tier predictions for ethyl (2E,4Z)-deca-dienoate were inconsistent across models, whereas targeted third-tier testing using the SENS-IS assay identified the compound as a strong sensitiser (GHS Category 1A). Overall, this study demonstrates how integrated modelling, Bayesian evidence updating and targeted NAM testing can reduce uncertainty in skin sensitization assessment. The SaferSkin framework provides a transparent and reproducible approach for implementing Next Generation Risk Assessment (NGRA) strategies and supports the development of animal-free regulatory toxicology and Safe-and-Sustainable-by-Design chemical innovation.