xTracer: Integrating chromatogram and mobiligram correlations for untargeted peptide identification in SLIM-based PAMAF data
xTracer: Integrating chromatogram and mobiligram correlations for untargeted peptide identification in SLIM-based PAMAF data
Song, J.; deng, l.; Royer, L.; Kalafut, B.; DeBord, D.; Meyer, J.
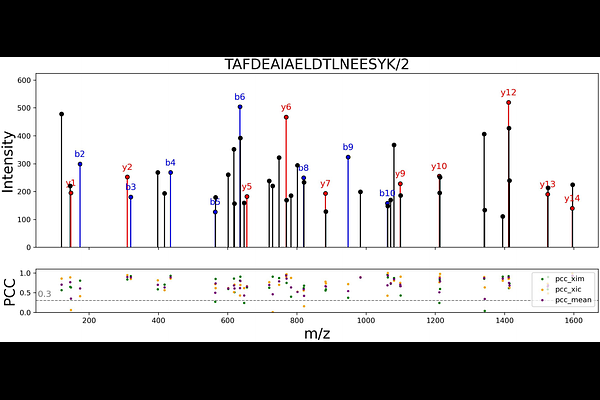
AbstractParallel Accumulation with Mobility Aligned Fragmentation (PAMAF) achieves near complete ion utilization and high spectral specificity by fragmenting all mobility separated precursors without quadrupole isolation. Leveraging the ultrahigh mobility resolution of SLIM, this quadrupole free strategy maximizes ion utilization efficiency and offers a promising approach in mass spectrometry based proteomics, particularly for low abundance peptides or low input samples. However, the unique data structure of PAMAF where precursor fragment relationships are encoded along the mobility dimension renders it incompatible with existing peptide identification tools. Here, we present xTracer, the first untargeted peptide identification algorithm developed specifically for PAMAF data. xTracer integrates correlations across both chromatographic and mobility dimensions to associate precursor and fragment ions, reconstruct pseudo-spectra, and enable database searching using well established DDA search engines. Applied to datasets with varying sample loads and acquisition throughputs, xTracer consistently achieved robust and reproducible peptide identifications, outperforming single domain correlation strategies. Overall, xTracer provides a versatile and high efficiency computational framework for reconstructing pseudo spectra from quadrupole free, mobility aligned fragmentation data, enhancing the analytical power of high resolution ion mobility (HRIM) based proteomics.