Substrate-dependent oligomerization modulates DGAT1 activity and subcellular localization
Substrate-dependent oligomerization modulates DGAT1 activity and subcellular localization
Sapia, J.; Salo, V.; Tasnim, A.; Campomanes, P.; Sui, X.; Vanni, S.
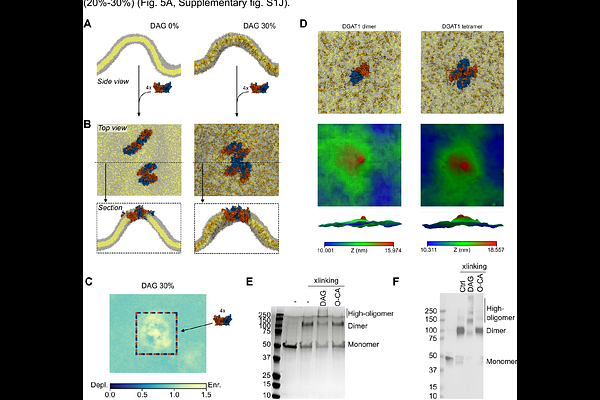
AbstractLipid Droplets (LDs) are ubiquitous organelles that are responsible for intracellular energy storage, in the form of highly esterified lipids such as triglycerides (TGs) and sterol esters. LDs emerge from and engage in stable contact sites with the endoplasmic reticulum (ER), where TG biosynthesis takes place by the action of the acyltransferase DGAT1. Despite the recent cryo-EM determination of the human dimeric structure of DGAT11,2, many aspects of the mechanism underlying TG synthesis in the ER remain unclear. Using a combination of molecular dynamics (MD) simulations, biochemical reconstitutions and fluorescence microscopy in live cells, we characterize several steps of DGAT1 molecular mechanism. We found that DAG preferentially enters the catalytic pocket of DGAT1 from the ER luminal leaflet, via a pathway that involves several conserved residues. Each DGAT1 subunit is able to bind multiple DAG molecules, and the presence of DAG promotes the formation of high-order DGAT1 oligomers. DGAT1 displays a preference for curved bilayers in silico, and it preferentially localizes in the ER tubular network, where LD formation is proposed to take place, upon the increase in its natural substrate diacylglycerol (DAG). Overall, our investigations provide a molecular view of how the interplay between protein oligomerization, subcellular localization and substrate biophysical properties modulate DGAT1 enzymatic activity.