Nar1 binds the cytosolic iron sulfur cluster assembly targeting complex via a bipartite interaction interface
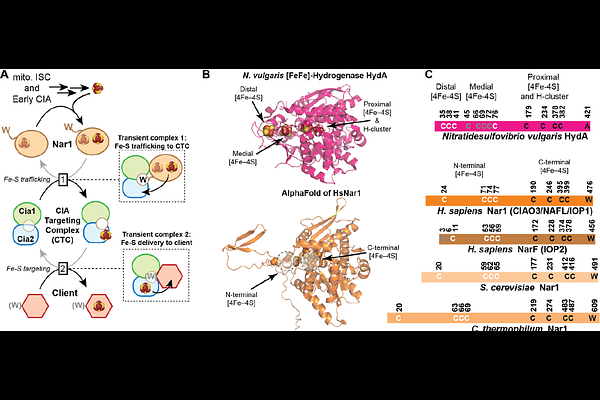
Nar1 binds the cytosolic iron sulfur cluster assembly targeting complex via a bipartite interaction interface
Buzuk, A.; Ho, J. V.; Marquez, M. D.; Wang, B.; Perlstein, D. L.
AbstractThe cytosolic iron-sulfur cluster assembly (CIA) pathway maturates essential nuclear and cytosolic Fe-S proteins required for genome maintenance and cellular metabolism. Nar1 (also called CIAO3 or IOP1) is a conserved Fe-S protein that connects the early and late steps of the CIA pathway, yet the molecular basis for its proposed function as a metallocluster carrier remains poorly defined. In particular, the interactions responsible for Nar1 recruitment to the CIA targeting complex (CTC) during cluster delivery remain unknown. Here, we define the molecular basis for Nar1 recruitment to the CTC using biochemical reconstitution, quantitative protein-protein interaction assays, and AlphaFold modeling. Our data reveal that Nar1 binds the CTC through two distinct interfaces. A primary interface comprises an electrostatic interaction that anchors Nar1 to a conserved acidic surface on the Cia1 subunit of the CTC and a secondary interface involves binding of Nar1's divergent targeting complex recognition peptide at the Cia1-Cia2 interface. Thus, Nar1 engages a conserved CTC surface that serves as a recruitment platform for multiple binding partners, including CIA clients. Computational structural models position the putative Fe-S cluster donor site of Nar1 adjacent to a proposed acceptor site on Cia2, suggesting that this bipartite binding mechanism positions Nar1 for transfer of an Fe-S cluster to the targeting complex. Together, these findings resolve conflicting models for Nar1 recruitment and establish a mechanistic framework for understanding how the CTC engages multiple binding partners during cytosolic iron-sulfur protein maturation.